The distribution of neurons expressing calcium-permeable AMPA receptors in the superficial laminae of the spinal cord dorsal horn
- PMID: 10066261
- PMCID: PMC6782571
- DOI: 10.1523/JNEUROSCI.19-06-02081.1999
The distribution of neurons expressing calcium-permeable AMPA receptors in the superficial laminae of the spinal cord dorsal horn
Abstract
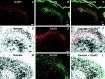
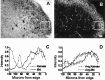
The superficial dorsal horn is a major site of termination of nociceptive primary afferents. Fast excitatory synaptic transmission in this region is mediated mainly by release of glutamate onto postsynaptic AMPA and NMDA receptors. NMDA receptors are known to be Ca2+-permeable and to provide synaptically localized Ca2+ signals that mediate short-term and long-term changes in synaptic strength. Less well known is a subpopulation of AMPA receptors that is Ca2+-permeable and has been shown to be synaptically localized on dorsal horn neurons in culture (Gu et al., 1996) and expressed by dorsal horn neurons in situ (Nagy et al., 1994; Engelman et al., 1997). We used kainate-induced cobalt uptake as a functional marker of neurons expressing Ca2+-permeable AMPA receptors and combined this with markers of nociceptive primary afferents in the postnatal rat dorsal horn. We have shown that cobalt-positive neurons are located in lamina I and outer lamina II, a region strongly innervated by nociceptors. These cobalt-positive neurons colocalize with afferents labeled by LD2, and with the most dorsal region of capsaicin-sensitive and IB4- and LA4-positive afferents. In contrast, inner lamina II has a sparser distribution of cobalt-positive neurons. Some lamina I neurons expressing the NK1 receptor, the receptor for substance P, are also cobalt positive. These neurons are likely to be projection neurons in the nociceptive pathway. On the basis of all of these observations, we propose that Ca2+-permeable AMPA receptors are localized to mediate transmission of nociceptive information.
Figures





References
-
- Albuquerque C, MacDermott AB. Selective expression of calcium-permeable AMPA receptors in subpopulations of inhibitory and excitatory dorsal horn neurons. Soc Neuroci Abstr. 1998;24:92.
-
- Bice TN, Beal JA. Quantitative and neurogenic analysis of the total population and subpopulations of neurons defined by axon projection in the superficial dorsal horn of the rat lumbar spinal cord. J Comp Neurol. 1997;288:550–564. - PubMed
-
- Burnashev N, Monyer H, Seeburg PH, Sakmann B. Divalent ion permeability of AMPA receptor channels is dominated by the edited form of a single subunit. Neuron. 1992;8:189–198. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
