Visualization of receptor-mediated endocytosis in yeast
- PMID: 10069819
- PMCID: PMC25203
- DOI: 10.1091/mbc.10.3.799
Visualization of receptor-mediated endocytosis in yeast
Abstract
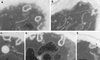
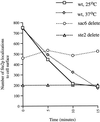
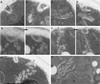
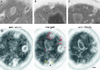
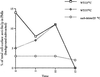
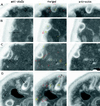
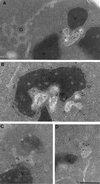
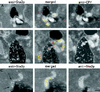
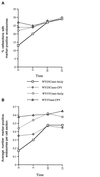
We studied the ligand-induced endocytosis of the yeast alpha-factor receptor Ste2p by immuno-electron microscopy. We observed and quantitated time-dependent loss of Ste2p from the plasma membrane of cells exposed to alpha-factor. This ligand-induced internalization of Ste2p was blocked in the well-characterized endocytosis-deficient mutant sac6Delta. We provide evidence that implicates furrow-like invaginations of the plasma membrane as the site of receptor internalization. These invaginations are distinct from the finger-like plasma membrane invaginations within actin cortical patches. Consistent with this, we show that Ste2p is not located within the cortical actin patch before and during receptor-mediated endocytosis. In wild-type cells exposed to alpha-factor we also observed and quantitated a time-dependent accumulation of Ste2p in intracellular, membrane-bound compartments. These compartments have a characteristic electron density but variable shape and size and are often located adjacent to the vacuole. In immuno-electron microscopy experiments these compartments labeled with antibodies directed against the rab5 homologue Ypt51p (Vps21p), the resident vacuolar protease carboxypeptidase Y, and the vacuolar H+-ATPase Vph1p. Using a new double-labeling technique we have colocalized antibodies against Ste2p and carboxypeptidase Y to this compartment, thereby identifying these compartments as prevacuolar late endosomes.
Figures
References
-
- Adams AE, Botstein D, Drubin D. Requirement of yeast fimbrin for actin organization and morphogenesis in vivo. Nature. 1991;354:404–408. - PubMed
-
- Anderson R, Vasile E, Mello R, Brown M, Goldstein J. Immunocytochemical visualization of coated pits and vesicles in human fibroblasts: relation to low density lipoprotein receptor distribution. Cell. 1978;15:919–933. - PubMed
-
- Botstein D, Amberg D, Huffaker T, Mulholland J, Adams A, Drubin D, Stearns T. The yeast cytoskeleton. In: Broach JR, Pringle JR, Jones EW, editors. The Molecular and Cellular Biology of the Yeast Saccharomyces. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory; 1997. pp. 1–90.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

