Antigen-stimulated dissociation of BCR mIg from Ig-alpha/Ig-beta: implications for receptor desensitization
- PMID: 10072076
- PMCID: PMC3931429
- DOI: 10.1016/s1074-7613(00)80024-2
Antigen-stimulated dissociation of BCR mIg from Ig-alpha/Ig-beta: implications for receptor desensitization
Abstract
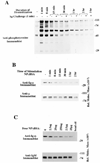
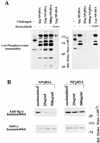
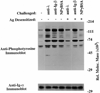
B cell antigen receptor (BCR) ligation leads to receptor desensitization wherein BCR remain competent to bind antigen and yet fail to transduce signals. Desensitized BCR exhibit a defect at the most proximal level of signal transduction, consistent with failed transmission of signals through the receptor complex. We report that antigen stimulation leads to dissociation or destabilization of the BCR reflected by inability to coimmunoprecipitate Ig-alpha/Ig-beta with mIg. This destabilization is temporally correlated with desensitization and occurs in BCR containing mIgM and mIgD. Induction of BCR destabilization requires tyrosine kinase activation but is not induced by phosphatase inhibitors. BCR destabilization occurs at the cell surface and "dissociated" Ig-alpha/Ig-beta complexes remain responsive to anti-Ig-beta stimulation, suggesting that mIg-transducer uncoupling may mediate receptor desensitization.
Figures




References
-
- Albrecht DL, Noelle RJ. Membrane Ig-cytoskeletal interactions. I. Flow cytofluorometric and biochemical analysis of membrane IgM-cytoskeletal interactions. J. Immunol. 1988;141:3915–3922. - PubMed
-
- Braun J, Hochman PS, Unanue ER. Ligand-induced association of surface immunoglobulin with the detergent-insoluble cytoskeletal matrix of the B lymphocyte. J. Immunol. 1982;128:1198–1204. - PubMed
-
- Brunswick M, June CH, Mond JJ. B lymphocyte immunoglobulin receptor desensitization is downstream of tyrosine kinase activation. Cell. Immunol. 1994;156:240–244. - PubMed
-
- Cambier JC. New nomenclature for the Reth motif (or ARH1/TAM/ARAM/YXXL) Immunol. Today. 1995;16:110. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

