Bacterial lipopolysaccharide inhibits dengue virus infection of primary human monocytes/macrophages by blockade of virus entry via a CD14-dependent mechanism
- PMID: 10074110
- PMCID: PMC104020
- DOI: 10.1128/JVI.73.4.2650-2657.1999
Bacterial lipopolysaccharide inhibits dengue virus infection of primary human monocytes/macrophages by blockade of virus entry via a CD14-dependent mechanism
Abstract
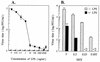
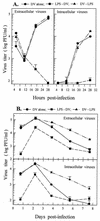
Monocytes/macrophages (MO/Mphi) are the major target cells for both dengue virus (DV) and bacterial lipopolysaccharide (LPS), and the aim of this study was to define their interactions. We had found that LPS markedly suppressed DV infection of primary human MO/Mphi when it was added to cultures prior to or together with, but not after, viral adsorption. The inhibitory effect of LPS was direct and specific and was not mediated by LPS-induced secretion of cytokines and chemokines such as tumor necrosis factor alpha, interleukin-1beta (IL-1beta), IL-6, IL-8, IL-12, alpha interferon, MIP-1alpha, and RANTES. In fact, productive DV infection was not blocked but was just postponed by LPS, with a time lag equal to one viral replication cycle. Time course studies demonstrated that LPS was only effective in suppressing DV infection of MO/Mphi that had not been previously exposed to the virus. At various time points after viral adsorption, the level of unbound viruses that remained free in the culture supernatants of LPS-pretreated cultures was much higher than that of untreated controls. These observations suggest that the LPS-induced suppression of DV replication was at the level of virus attachment and/or entry. Blockade of the major LPS receptor, CD14, with monoclonal antibodies MY4 or MoS39 failed to inhibit DV infection but could totally abrogate the inhibitory effect of LPS. Moreover, human serum could significantly enhance the LPS-induced DV suppression in a CD14-dependent manner, indicating that the "binding" of LPS to CD14 was critical for the induction of virus inhibition. Taken together, our results suggest that LPS blocked DV entry into human MO/Mphi via its receptor CD14 and that a CD14-associated cell surface structure may be essential for the initiation of a DV infection.
Figures

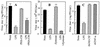


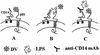
References
-
- Bergelson J M, Cunningham J A, Droguett G, Kurt-Jones E A, Krithivas A, Hong J S, Horwitz M S, Crowell R L, Finberg R W. Isolation of a common receptor for coxsackie B viruses and adenoviruses 2 and 5. Science. 1997;275:1320–1323. - PubMed
-
- Biswas P, Poli G, Kinter A L, Justement J S, Stanley S K, Maury W J, Bressler P J M, Orenstein J M, Fauci A S. Interferon γ modulates the expression of human immunodeficiency virus in persistently infected promonocytic cells by redirecting the production of virions to intracytoplasmic vacuoles. J Exp Med. 1992;176:739–750. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

