Expression of specific chemokines and chemokine receptors in the central nervous system of multiple sclerosis patients
- PMID: 10079101
- PMCID: PMC408141
- DOI: 10.1172/JCI5150
Expression of specific chemokines and chemokine receptors in the central nervous system of multiple sclerosis patients
Abstract
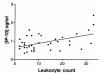
Chemokines direct tissue invasion by specific leukocyte populations. Thus, chemokines may play a role in multiple sclerosis (MS), an idiopathic disorder in which the central nervous system (CNS) inflammatory reaction is largely restricted to mononuclear phagocytes and T cells. We asked whether specific chemokines were expressed in the CNS during acute demyelinating events by analyzing cerebrospinal fluid (CSF), whose composition reflects the CNS extracellular space. During MS attacks, we found elevated CSF levels of three chemokines that act toward T cells and mononuclear phagocytes: interferon-gamma-inducible protein of 10 kDa (IP-10); monokine induced by interferon-gamma (Mig); and regulated on activation, normal T-cell expressed and secreted (RANTES). We then investigated whether specific chemokine receptors were expressed by infiltrating cells in demyelinating MS brain lesions and in CSF. CXCR3, an IP-10/Mig receptor, was expressed on lymphocytic cells in virtually every perivascular inflammatory infiltrate in active MS lesions. CCR5, a RANTES receptor, was detected on lymphocytic cells, macrophages, and microglia in actively demyelinating MS brain lesions. Compared with circulating T cells, CSF T cells were significantly enriched for cells expressing CXCR3 or CCR5. Our results imply pathogenic roles for specific chemokine-chemokine receptor interactions in MS and suggest new molecular targets for therapeutic intervention.
Figures



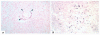
References
-
- Trapp B, et al. Axonal transection in the lesions of multiple sclerosis. N Engl J Med. 1998;338:278–290. - PubMed
-
- Rudick R, Cohen J, Weinstock-Guttman B, Kinkel R, Ransohoff R. Management of multiple sclerosis. N Engl J Med. 1996;337:1604–1612. - PubMed
-
- Sørensen T, Ransohoff R. Etiology and pathogenesis of multiple sclerosis. Semin Neurol. 1998;18:287–295. - PubMed
-
- Steinman L. Multiple sclerosis: a coordinated immunological attack against myelin in the central nervous system. Cell. 1996;85:299–302. - PubMed
-
- Rudick R, et al. Cerebrospinal fluid abnormalities in a phase III trial of avonex (IFNB-1a) for relapsing multiple sclerosis. J Neuroimmunol. 1999;93:8–15. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

