Defensins impair phagocytic killing by neutrophils in biomaterial-related infection
- PMID: 10084997
- PMCID: PMC96507
- DOI: 10.1128/IAI.67.4.1640-1645.1999
Defensins impair phagocytic killing by neutrophils in biomaterial-related infection
Abstract
The implantation of foreign material carries a risk of infection which frequently is resistant to all treatment short of removing the implant. We have previously shown that these materials activate neutrophils by contact, leading to production of oxygen free radicals accompanied by release of granule products. Such activation further results in depletion of local host defenses, including the capacity of biomaterial-activated neutrophils to kill bacteria. Among the granule products released from neutrophils are small cationic antibacterial peptides (human neutrophil peptides [HNP]) known as defensins. Here we tested the hypothesis that defensins, released from activated neutrophils onto the surface of biomaterials, might play a role in the deactivation of subsequent neutrophil populations. Incubation of neutrophils with purified HNP resulted in a dose-related impairment of stimulus-induced oxygen radical production and of phagocytic killing. Furthermore, fresh neutrophils added to biomaterial-associated neutrophils exhibited impaired phagocytic killing. This impairment could be abrogated by antibody to HNP but not by an irrelevant antibody. Taken together, these observations support the idea that neutrophils activated at a material surface can create, by means of HNP release, an environment hostile to their microbicidal function and that of their infiltrating brethren.
Figures

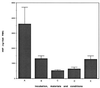


 ) and 25
(░⃞) μg of HNP1/ml had little effect (mean of two experiments).
) and 25
(░⃞) μg of HNP1/ml had little effect (mean of two experiments).
 ),
but this is overcome by addition of monoclonal antibody specific for
HNP (░⃞) but not by irrelevant antibody to VCAM (▨) prior to the
addition of fresh neutrophils. This effect was highly significant at
both 2 and 24 h after the inoculation of neutrophils with
staphylococci (P = 0.00008).
),
but this is overcome by addition of monoclonal antibody specific for
HNP (░⃞) but not by irrelevant antibody to VCAM (▨) prior to the
addition of fresh neutrophils. This effect was highly significant at
both 2 and 24 h after the inoculation of neutrophils with
staphylococci (P = 0.00008).Similar articles
-
In vitro antibacterial activities of platelet microbicidal protein and neutrophil defensin against Staphylococcus aureus are influenced by antibiotics differing in mechanism of action.Antimicrob Agents Chemother. 1999 May;43(5):1111-7. doi: 10.1128/AAC.43.5.1111. Antimicrob Agents Chemother. 1999. PMID: 10223922 Free PMC article.
-
Effect of plasma and matrix proteins on defensin-induced impairment of phagocytic killing by adherent neutrophils.J Biomed Mater Res. 2001 Oct;57(1):1-7. doi: 10.1002/1097-4636(200110)57:1<1::aid-jbm1134>3.0.co;2-r. J Biomed Mater Res. 2001. PMID: 11416842
-
Comparative studies on the extracellular release and biological activity of guinea pig neutrophil cationic antibacterial polypeptide of 11 kDa (CAP11) and defensins.Comp Biochem Physiol B Biochem Mol Biol. 1997 Jan;116(1):99-107. doi: 10.1016/s0305-0491(96)00222-2. Comp Biochem Physiol B Biochem Mol Biol. 1997. PMID: 9080667
-
Epic Immune Battles of History: Neutrophils vs. Staphylococcus aureus.Front Cell Infect Microbiol. 2017 Jun 30;7:286. doi: 10.3389/fcimb.2017.00286. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 28713774 Free PMC article. Review.
-
Neutrophil antibacterial peptides, multifunctional effector molecules in the mammalian immune system.J Immunol Methods. 1999 Dec 17;232(1-2):45-54. doi: 10.1016/s0022-1759(99)00152-0. J Immunol Methods. 1999. PMID: 10618508 Review.
Cited by
-
Interleukin-1 receptor type I gene-deficient mice are less susceptible to Staphylococcus epidermidis biomaterial-associated infection than are wild-type mice.Infect Immun. 2000 Dec;68(12):6924-31. doi: 10.1128/IAI.68.12.6924-6931.2000. Infect Immun. 2000. PMID: 11083815 Free PMC article.
-
In vitro interactions between bacteria, osteoblast-like cells and macrophages in the pathogenesis of biomaterial-associated infections.PLoS One. 2011;6(9):e24827. doi: 10.1371/journal.pone.0024827. Epub 2011 Sep 13. PLoS One. 2011. PMID: 21931858 Free PMC article.
-
Granulocyte colony-stimulating factor for the treatment of biomaterial-associated staphylococcal infections in-vitro.Urol Res. 2003 Feb;30(6):394-8. doi: 10.1007/s00240-002-0289-7. Epub 2002 Dec 20. Urol Res. 2003. PMID: 12599021
-
Free Radical Production in Immune Cell Systems Induced by Ti, Ti6Al4V and SS Assessed by Chemiluminescence Probe Pholasin Assay.Int J Biomater. 2012;2012:380845. doi: 10.1155/2012/380845. Epub 2012 Jun 19. Int J Biomater. 2012. Retraction in: Int J Biomater. 2013;2013:148185. doi: 10.1155/2013/148185. PMID: 22778739 Free PMC article. Retracted.
-
Direct Microscopic Observation of Human Neutrophil-Staphylococcus aureus Interaction In Vitro Suggests a Potential Mechanism for Initiation of Biofilm Infection on an Implanted Medical Device.Infect Immun. 2019 Nov 18;87(12):e00745-19. doi: 10.1128/IAI.00745-19. Print 2019 Dec. Infect Immun. 2019. PMID: 31548325 Free PMC article.
References
-
- Arizono T, Oga M, Sugioka Y. Increased resistance of bacteria after adherence to polymethyl methacrylate. Acta Orthop Scand. 1992;63:661–664. - PubMed
-
- Babior B M. Oxidants from phagocytes: agents of defense and destruction. Blood. 1984;64:959–966. - PubMed
-
- Baddour L M, Christensen G D, Hester M G, Bisno A L. Production of experimental endocarditis by coagulase-negative staphylococci: variability in species virulence. J Infect Dis. 1984;150:721–727. - PubMed
-
- Borregaard N, Lollike K, Kyeldsen L, Sengelov H, Bastholm L, Nielsen M H, Bainton D F. Human neutrophil granules and secretory vesicles. Eur J Haematol. 1993;51:187–198. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

