SNARE membrane trafficking dynamics in vivo
- PMID: 10085287
- PMCID: PMC2148207
- DOI: 10.1083/jcb.144.5.869
SNARE membrane trafficking dynamics in vivo
Abstract
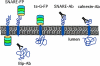
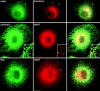
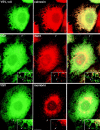
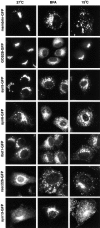
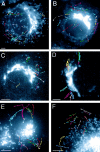
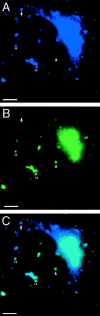
The ER/Golgi soluble NSF attachment protein receptor (SNARE) membrin, rsec22b, and rbet1 are enriched in approximately 1-micrometer cytoplasmic structures that lie very close to the ER. These appear to be ER exit sites since secretory cargo concentrates in and exits from these structures. rsec22b and rbet1 fused to fluorescent proteins are enriched at approximately 1-micrometer ER exit sites that remained more or less stationary, but periodically emitted streaks of fluorescence that traveled generally in the direction of the Golgi complex. These exit sites were reused and subsequent tubules or streams of vesicles followed similar trajectories. Fluorescent membrin- enriched approximately 1-micrometer peripheral structures were more mobile and appeared to translocate through the cytoplasm back and forth, between the periphery and the Golgi area. These mobile structures could serve to collect secretory cargo by fusing with ER-derived vesicles and ferrying the cargo to the Golgi. The post-Golgi SNAREs, syntaxin 6 and syntaxin 13, when fused to fluorescent proteins each displayed characteristic patterns of movement. However, syntaxin 13 was the only SNARE whose life cycle appeared to involve interactions with the plasma membrane. These studies reveal the in vivo spatiotemporal dynamics of SNARE proteins and provide new insight into their roles in membrane trafficking.
Figures




References
-
- Advani RJ, Bae HR, Bock JB, Chao DS, Doung YC, Prekeris R, Yoo JS, Scheller RH. Seven novel mammalian SNARE proteins localize to distinct membrane compartments. J Biol Chem. 1998;273:10317–10324. - PubMed
-
- Balch WE, McCafferey JM, Plunter H, Farquhar MG. Vesicular stomatitis virus glycoprotein is sorted and concentrated during export from the endoplasmic reticulum. Cell. 1994;76:841–852. - PubMed
-
- Bergmann JE. Using temperature-sensitive mutants of VSV to study membrane protein biogenesis. Methods Cell Biol. 1989;32:85–110. - PubMed

