ATP counteracts the rundown of gap junctional channels of rat ventricular myocytes by promoting protein phosphorylation
- PMID: 10087344
- PMCID: PMC2269282
- DOI: 10.1111/j.1469-7793.1999.0447v.x
ATP counteracts the rundown of gap junctional channels of rat ventricular myocytes by promoting protein phosphorylation
Abstract
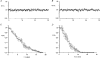
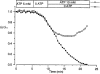
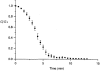
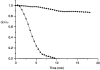
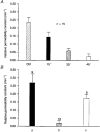
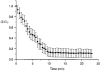
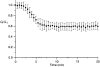
1. The degree of cell-to-cell coupling between ventricular myocytes of neonatal rats appeared well preserved when studied in the perforated version of the patch clamp technique or, in double whole-cell conditions, when ATP was present in the patch pipette solution. In contrast, when ATP was omitted, the amplitude of junctional current rapidly declined (rundown). 2. To examine the mechanism(s) of ATP action, an 'internal perfusion technique' was adapted to dual patch clamp conditions, and reintroduction of ATP partially reversed the rundown of junctional channels. 3. Cell-to-cell communication was not preserved by a non-hydrolysable ATP analogue (5'-adenylimidodiphosphate, AMP-PNP), indicating that the effect most probably did not involve direct interaction of ATP with the channel-forming proteins. 4. An ATP analogue supporting protein phosphorylation but not active transport processes (adenosine 5'-O-(3-thiotriphosphate), ATPgammaS) maintained normal intercellular communication, suggesting that the effect was due to kinase activity rather than to altered intracellular Ca2+. 5. A broad spectrum inhibitor of endogenous serine/threonine protein kinases (H7) reversibly reduced the intercellular coupling. A non-specific exogenous protein phosphatase (alkaline phosphatase) mimicked the effects of ATP deprivation. The non-specific inhibition of endogenous protein phosphatases resulted in the preservation of substantial cell-to-cell communication in ATP-free conditions. 6. The activity of gap junctional channels appears to require both the presence of ATP and protein kinase activity to counteract the tonic activity of endogenous phosphatase(s).
Figures


References
-
- Becq F. Ionic channel rundown in excised membrane patches. Biochimica et Biophysica Acta. 1996;1286:53–63. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

