Crystal structure of human type II inosine monophosphate dehydrogenase: implications for ligand binding and drug design
- PMID: 10097070
- PMCID: PMC22327
- DOI: 10.1073/pnas.96.7.3531
Crystal structure of human type II inosine monophosphate dehydrogenase: implications for ligand binding and drug design
Abstract
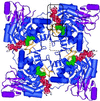
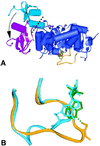
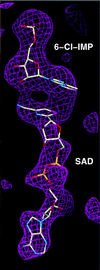
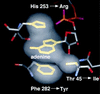
Inosine monophosphate dehydrogenase (IMPDH) controls a key metabolic step in the regulation of cell growth and differentiation. This step is the NAD-dependent oxidation of inosine 5' monophosphate (IMP) to xanthosine 5' monophosphate, the rate-limiting step in the synthesis of the guanine nucleotides. Two isoforms of IMPDH have been identified, one of which (type II) is significantly up- regulated in neoplastic and differentiating cells. As such, it has been identified as a major target in antitumor and immunosuppressive drug design. We present here the 2.9-A structure of a ternary complex of the human type II isoform of IMPDH. The complex contains the substrate analogue 6-chloropurine riboside 5'-monophosphate (6-Cl-IMP) and the NAD analogue selenazole-4-carboxamide adenine dinucleotide, the selenium derivative of the active metabolite of the antitumor drug tiazofurin. The enzyme forms a homotetramer, with the dinucleotide binding at the monomer-monomer interface. The 6 chloro-substituted purine base is dehalogenated, forming a covalent adduct at C6 with Cys-331. The dinucleotide selenazole base is stacked against the 6-Cl-IMP purine ring in an orientation consistent with the B-side stereochemistry of hydride transfer seen with NAD. The adenosine end of the ligand interacts with residues not conserved between the type I and type II isoforms, suggesting strategies for the design of isoform-specific agents.
Figures
References
-
- Weber G, Prajda N, Abonyi M, Look K Y, Tricot G. Anticancer Res. 1996;16:3313–3322. - PubMed
-
- Wu J C. Perspect Drug Discovery Des. 1994;2:185–204.
-
- Franklin T J, Edwards G, Hedge P. Adv Exp Med Biol. 1994;370:155–160. - PubMed
-
- Weber G, Prajda N, Jackson R C. Adv Enzyme Regul. 1976;14:3–24. - PubMed
-
- Jackson R C, Weber G, Morris H P. Nature (London) 1975;256:331–333. - PubMed
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

