Ectopic expression of the minimal whiE polyketide synthase generates a library of aromatic polyketides of diverse sizes and shapes
- PMID: 10097087
- PMCID: PMC22344
- DOI: 10.1073/pnas.96.7.3622
Ectopic expression of the minimal whiE polyketide synthase generates a library of aromatic polyketides of diverse sizes and shapes
Abstract
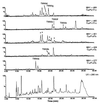
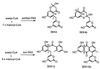
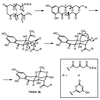
The single recombinant expressing the Streptomyces coelicolor minimal whiE (spore pigment) polyketide synthase (PKS) is uniquely capable of generating a large array of well more than 30 polyketides, many of which, so far, are novel to this recombinant. The characterized polyketides represent a diverse set of molecules that differ in size (chain length) and shape (cyclization pattern). This combinatorial biosynthetic library is, by far, the largest and most complex of its kind described to date and indicates that the minimal whiE PKS does not independently control polyketide chain length nor dictate the first cyclization event. Rather, the minimal PKS enzyme complex must rely on the stabilizing effects of additional subunits (i.e., the cyclase whiE-ORFVI) to ensure that the chain reaches the full 24 carbons and cyclizes correctly. This dramatic loss of control implies that the growing polyketide chain does not remain enzyme bound, resulting in the spontaneous cyclization of the methyl terminus. Among the six characterized dodecaketides, four different first-ring cyclization regiochemistries are represented, including C7/C12, C8/C13, C10/C15, and C13/C15. The dodecaketide TW93h possesses a unique 2,4-dioxaadamantane ring system and represents a new structural class of polyketides with no related structures isolated from natural or engineered organisms, thus supporting the claim that engineered biosynthesis is capable of producing novel chemotypes.
Figures


Comment in
-
Microbial polyketide synthases: more and more prolific.Proc Natl Acad Sci U S A. 1999 Mar 30;96(7):3336-8. doi: 10.1073/pnas.96.7.3336. Proc Natl Acad Sci U S A. 1999. PMID: 10097038 Free PMC article. Review. No abstract available.
References
-
- Fu H, Khosla C. Mol Diversity. 1995;1:121–124. - PubMed
-
- Tsoi C J, Khosla C. Chem Biol. 1995;2:355–362. - PubMed
-
- O’Hagan D. The Polyketide Metabolites. Chichester, U.K.: Ellis Horwood; 1991.
-
- Hopwood D A. Chem Rev. 1997;97:2465–2497. - PubMed
-
- Cortes J, Haydock S F, Roberts G A, Bevitt D J, Leadlay P F. Nature (London) 1990;348:176–178. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

