Disruption of estrogen signaling does not prevent progesterone action in the estrogen receptor alpha knockout mouse uterus
- PMID: 10097091
- PMCID: PMC22348
- DOI: 10.1073/pnas.96.7.3646
Disruption of estrogen signaling does not prevent progesterone action in the estrogen receptor alpha knockout mouse uterus
Abstract
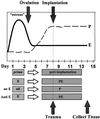
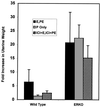
Estrogen is known to increase progesterone receptor (PR) levels in the wild-type mouse uterus, and this estrogen induction was thought to be important for progesterone action through the PR. The estrogen receptor alpha knockout (ERKO) mouse uterus was observed to express PR mRNA that cannot be induced by estrogen. Progesterone action was characterized to determine whether it was diminished in ERKO mice. The PR protein is present in the ERKO uterus at 60% of the level measured in a wild-type uterus. The PR-A and PR-B isoforms are both detected on Western blot, and the ratio of isoforms is the same in both genotypes. Although the level of PR is reduced in the ERKO uterus, the receptor level is sufficient to induce genomic responses, since both calcitonin and amphiregulin mRNAs were increased after progesterone treatment. Finally, the ERKO uterus can be induced to undergo a progesterone-dependent decidual response. Surprisingly, the decidual response is estrogen independent in the ERKO, although it remains estrogen dependent in a wild type. These results indicate that estrogen receptor alpha modulation of PR levels is not necessary for expression of the PR or genomic and physiologic responses to progesterone in the ERKO uterus.
Figures




Similar articles
-
Analysis of transcription and estrogen insensitivity in the female mouse after targeted disruption of the estrogen receptor gene.Mol Endocrinol. 1995 Nov;9(11):1441-54. doi: 10.1210/mend.9.11.8584021. Mol Endocrinol. 1995. PMID: 8584021
-
Regulation of progesterone receptors and decidualization in uterine stroma of the estrogen receptor-alpha knockout mouse.Biol Reprod. 2001 Jan;64(1):272-83. doi: 10.1095/biolreprod64.1.272. Biol Reprod. 2001. PMID: 11133684
-
Studies using the estrogen receptor alpha knockout uterus demonstrate that implantation but not decidualization-associated signaling is estrogen dependent.Biol Reprod. 2002 Oct;67(4):1268-77. doi: 10.1095/biolreprod67.4.1268. Biol Reprod. 2002. PMID: 12297545
-
Progesterone action and responses in the alphaERKO mouse.Steroids. 2000 Oct-Nov;65(10-11):551-7. doi: 10.1016/s0039-128x(00)00113-6. Steroids. 2000. PMID: 11108859 Review.
-
Estrogen receptor null mice: what have we learned and where will they lead us?Endocr Rev. 1999 Jun;20(3):358-417. doi: 10.1210/edrv.20.3.0370. Endocr Rev. 1999. PMID: 10368776 Review.
Cited by
-
Perfluorooctanoic acid effects on ovaries mediate its inhibition of peripubertal mammary gland development in Balb/c and C57Bl/6 mice.Reprod Toxicol. 2012 Jul;33(4):563-576. doi: 10.1016/j.reprotox.2012.02.004. Epub 2012 Mar 5. Reprod Toxicol. 2012. PMID: 22414604 Free PMC article.
-
Molecular mechanisms of estrogen action in female genital tract development.Differentiation. 2021 Mar-Apr;118:34-40. doi: 10.1016/j.diff.2021.01.002. Epub 2021 Feb 24. Differentiation. 2021. PMID: 33707128 Free PMC article.
-
Estrogen receptors: structure, mechanisms and function.Rev Endocr Metab Disord. 2002 Sep;3(3):193-200. doi: 10.1023/a:1020068224909. Rev Endocr Metab Disord. 2002. PMID: 12215714 Review. No abstract available.
-
Progesterone receptors: form and function in brain.Front Neuroendocrinol. 2008 May;29(2):313-39. doi: 10.1016/j.yfrne.2008.02.001. Epub 2008 Feb 23. Front Neuroendocrinol. 2008. PMID: 18374402 Free PMC article. Review.
-
Estrogen Receptor Function: Impact on the Human Endometrium.Front Endocrinol (Lausanne). 2022 Feb 28;13:827724. doi: 10.3389/fendo.2022.827724. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35295981 Free PMC article. Review.
References
-
- Weitlauf H M. In: The Physiology of Reproduction. Knobil E, Neill J, editors. New York: Raven; 1988. pp. 231–262.
-
- Graham J D, Clarke C L. Endocr Rev. 1997;18:502–519. - PubMed
-
- Couse J F, Curtis S W, Washburn T F, Lindzey J, Golding T S, Lubahn D B, Smithies O, Korach K S. Mol Endocrinol. 1995;9:1441–1454. - PubMed
-
- Lydon J P, DeMayo F J, Funk C R, Mani S K, Hughes A R, Montgomery C A, Shyamala G, Conneely O M, O’Malley B W. Genes Dev. 1995;9:2266–2278. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

