Specific association of the gene product of PKD2 with the TRPC1 channel
- PMID: 10097141
- PMCID: PMC22398
- DOI: 10.1073/pnas.96.7.3934
Specific association of the gene product of PKD2 with the TRPC1 channel
Abstract
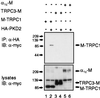
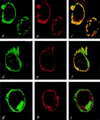
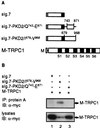
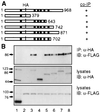
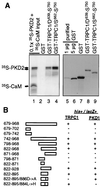
The function(s) of the genes (PKD1 and PKD2) responsible for the majority of cases of autosomal dominant polycystic kidney disease is unknown. While PKD1 encodes a large integral membrane protein containing several structural motifs found in known proteins involved in cell-cell or cell-matrix interactions, PKD2 has homology to PKD1 and the major subunit of the voltage-activated Ca2+ channels. We now describe sequence homology between PKD2 and various members of the mammalian transient receptor potential channel (TRPC) proteins, thought to be activated by G protein-coupled receptor activation and/or depletion of internal Ca2+ stores. We show that PKD2 can directly associate with TRPC1 but not TRPC3 in transfected cells and in vitro. This association is mediated by two distinct domains in PKD2. One domain involves a minimal region of 73 amino acids in the C-terminal cytoplasmic tail of PKD2 shown previously to constitute an interacting domain with PKD1. However, distinct residues within this region mediate specific interactions with TRPC1 or PKD1. The C-terminal domain is sufficient but not necessary for the PKD2-TRPC1 association. A more N-terminal domain located within transmembrane segments S2 and S5, including a putative pore helical region between S5 and S6, is also responsible for the association. Given the ability of the TRPC to form functional homo- and heteromultimeric complexes, these data provide evidence that PKD2 may be functionally related to TRPC proteins and suggest a possible role of PKD2 in modulating Ca2+ entry in response to G protein-coupled receptor activation and/or store depletion.
Figures

References
-
- Gabow P A. N Engl J Med. 1993;329:332–342. - PubMed
-
- The European Polycystic Kidney Disease Consortium. Cell. 1994;77:881–894. - PubMed
-
- The International Polycystic Kidney Disease Consortium. Cell. 1995;81:289–298. - PubMed
-
- Mochizuki T, Wu G, Hayashi T, Xenophontos S L, Veldhuisen B, Saris J J, Reynolds D M, Cai Y, Gabow P A, Pierides A, et al. Science. 1996;272:1339–1342. - PubMed
-
- Sandford R, Sgotto B, Aparicio S, Brenner S, Vaudin M, Wilson R K, Chissoe S, Pepin K, Bateman A, Chothia C, Hughes J, Harris P. Hum Mol Genet. 1997;6:1483–1489. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

