Deficiency of platelet-activating factor acetylhydrolase is a severity factor for asthma
- PMID: 10194471
- PMCID: PMC408260
- DOI: 10.1172/JCI5574
Deficiency of platelet-activating factor acetylhydrolase is a severity factor for asthma
Abstract
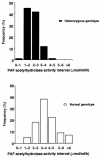
Asthma, a family of airway disorders characterized by airway inflammation, has an increasing incidence worldwide. Platelet-activating factor (PAF) may play a role in the pathophysiology of asthma. Its proinflammatory actions are antagonized by PAF acetylhydrolase. A missense mutation (V279F) in the PAF acetylhydrolase gene results in the complete loss of activity, which occurs in 4% of the Japanese population. We asked if PAF acetylhydrolase deficiency correlates with the incidence and severity of asthma in Japan. We found that the prevalence of PAF acetylhydrolase deficiency is higher in Japanese asthmatics than healthy subjects and that the severity of this syndrome is highest in homozygous-deficient subjects. We conclude that the PAF acetylhydrolase gene is a modulating locus for the severity of asthma.
Figures







References
-
- Lemanske RF, Jr, Busse WW. Asthma. JAMA. 1997;278:1855–1873. - PubMed
-
- Drazen JM. New directions in asthma drug therapy. Hosp Pract. 1998;33:25–26. - PubMed
-
- Davies RJ, et al. New insights into the understanding of asthma. Chest. 1997;111:2S–10S. - PubMed
-
- Rachelefsky G. Childhood asthma and allergic rhinitis: the role of leukotrienes. J Pediatr. 1997;131:348–355. - PubMed
-
- Page CP. The role of platelet-activating factor in asthma. J Allergy Clin Immunol. 1988;81:44–152. - PubMed

