Acarbose, a pseudooligosaccharide, is transported but not metabolized by the maltose-maltodextrin system of Escherichia coli
- PMID: 10198028
- PMCID: PMC93690
- DOI: 10.1128/JB.181.8.2612-2619.1999
Acarbose, a pseudooligosaccharide, is transported but not metabolized by the maltose-maltodextrin system of Escherichia coli
Abstract
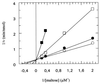
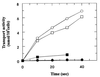
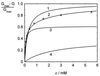
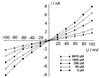
The pseudooligosaccharide acarbose is a potent inhibitor of amylases, glucosidases, and cyclodextrin glycosyltransferase and is clinically used for the treatment of so-called type II or insulin-independent diabetes. The compound consists of an unsaturated aminocyclitol, a deoxyhexose, and a maltose. The unsaturated aminocyclitol moiety (also called valienamine) is primarily responsible for the inhibition of glucosidases. Due to its structural similarity to maltotetraose, we have investigated whether acarbose is recognized as a substrate by the maltose/maltodextrin system of Escherichia coli. Acarbose at millimolar concentrations specifically affected the growth of E. coli K-12 on maltose as the sole source of carbon and energy. Uptake of radiolabeled maltose was competitively inhibited by acarbose, with a Ki of 1.1 microM. Maltose-grown cells transported radiolabeled acarbose, indicating that the compound is recognized as a substrate. Studying the interaction of acarbose with purified maltoporin in black lipid membranes revealed that the kinetics of acarbose binding to LamB is asymmetric. The on-rate of acarbose is approximately 30 times lower when the molecule enters the pore from the extracellular side than when it enters from the periplasmic side. Acarbose could not be utilized as a carbon source since the compound alone was not a substrate of amylomaltase (MalQ) and was only poorly attacked by maltodextrin glucosidase (MalZ).
Figures




References
-
- Al Kazaz M, Desseaux V, Marchis-Mouren G, Prodanov E, Santimone M. The mechanism of porcine pancreatic α-amylase. Inhibition of maltopentaose hydrolysis by acarbose, maltose and maltotriose. Eur J Biochem. 1998;252:100–107. - PubMed
-
- Benz R, Schmid A, Vos-Scheperkeuter G H. Mechanism of sugar transport through the sugar-specific LamB channel of Escherichia coli outer membrane. J Membr Biol. 1987;100:12–29. - PubMed
-
- Benz R, Janko K, Boos W, Läuger P. Formation of large, ion-permeable membrane channels by the matrix protein (porin) of Escherichia coli. Biochim Biophys Acta. 1978;511:305–319. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

