Expression of the antiproliferative gene TIS21 at the onset of neurogenesis identifies single neuroepithelial cells that switch from proliferative to neuron-generating division
- PMID: 10200315
- PMCID: PMC16385
- DOI: 10.1073/pnas.96.8.4639
Expression of the antiproliferative gene TIS21 at the onset of neurogenesis identifies single neuroepithelial cells that switch from proliferative to neuron-generating division
Abstract
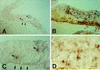
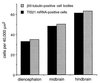
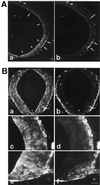
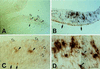
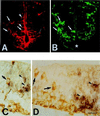
At the onset of mammalian neurogenesis, neuroepithelial (NE) cells switch from proliferative to neuron-generating divisions. Understanding the molecular basis of this switch requires the ability to distinguish between these two types of division. Here we show that in the mouse ventricular zone, expression of the mRNA of the antiproliferative gene TIS21 (PC3, BTG2) (i) starts at the onset of neurogenesis, (ii) is confined to a subpopulation of NE cells that increases in correlation with the progression of neurogenesis, and (iii) is not detected in newborn neurons. Expression of the TIS21 mRNA in the NE cells occurs transiently during the cell cycle, i.e., in the G1 phase. In contrast to the TIS21 mRNA, the TIS21 protein persists through the division of NE cells and is inherited by the neurons, where it remains detectable during neuronal migration and the initial phase of differentiation. Our observations indicate that the TIS21 gene is specifically expressed in those NE cells that, at their next division, will generate postmitotic neurons, but not in proliferating NE cells. Using TIS21 as a marker, we find that the switch from proliferative to neuron-generating divisions is initiated in single NE cells rather than in synchronized neighboring cells.
Figures



Similar articles
-
The gene PC3(TIS21/BTG2), prototype member of the PC3/BTG/TOB family: regulator in control of cell growth, differentiation, and DNA repair?J Cell Physiol. 2001 May;187(2):155-65. doi: 10.1002/jcp.1062. J Cell Physiol. 2001. PMID: 11267995 Review.
-
Expression of B-cell translocation gene 2 protein in normal human tissues.Tissue Cell. 2002 Feb;34(1):28-32. doi: 10.1054/tice.2001.0220. Tissue Cell. 2002. PMID: 11989967
-
BTG2(TIS21/PC3) induces neuronal differentiation and prevents apoptosis of terminally differentiated PC12 cells.Oncogene. 2002 Oct 3;21(44):6772-78. doi: 10.1038/sj.onc.1205888. Oncogene. 2002. PMID: 12360398
-
Selective lengthening of the cell cycle in the neurogenic subpopulation of neural progenitor cells during mouse brain development.J Neurosci. 2005 Jul 13;25(28):6533-8. doi: 10.1523/JNEUROSCI.0778-05.2005. J Neurosci. 2005. PMID: 16014714 Free PMC article.
-
Control of the Normal and Pathological Development of Neural Stem and Progenitor Cells by the PC3/Tis21/Btg2 and Btg1 Genes.J Cell Physiol. 2015 Dec;230(12):2881-90. doi: 10.1002/jcp.25038. J Cell Physiol. 2015. PMID: 25967096 Review.
Cited by
-
A novel function of DELTA-NOTCH signalling mediates the transition from proliferation to neurogenesis in neural progenitor cells.PLoS One. 2007 Nov 14;2(11):e1169. doi: 10.1371/journal.pone.0001169. PLoS One. 2007. PMID: 18000541 Free PMC article.
-
Ephrin B1 is expressed on neuroepithelial cells in correlation with neocortical neurogenesis.J Neurosci. 2001 Apr 15;21(8):2726-37. doi: 10.1523/JNEUROSCI.21-08-02726.2001. J Neurosci. 2001. PMID: 11306625 Free PMC article.
-
Forced G1-phase reduction alters mode of division, neuron number, and laminar phenotype in the cerebral cortex.Proc Natl Acad Sci U S A. 2009 Dec 22;106(51):21924-9. doi: 10.1073/pnas.0909894106. Epub 2009 Dec 3. Proc Natl Acad Sci U S A. 2009. PMID: 19959663 Free PMC article.
-
Neurons derive from the more apical daughter in asymmetric divisions in the zebrafish neural tube.Nat Neurosci. 2010 Jun;13(6):673-9. doi: 10.1038/nn.2547. Epub 2010 May 9. Nat Neurosci. 2010. PMID: 20453852
-
Single-cell analysis reveals transcriptional heterogeneity of neural progenitors in human cortex.Nat Neurosci. 2015 May;18(5):637-46. doi: 10.1038/nn.3980. Epub 2015 Mar 3. Nat Neurosci. 2015. PMID: 25734491 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

