Stable expression of human beta1,4-galactosyltransferase in plant cells modifies N-linked glycosylation patterns
- PMID: 10200324
- PMCID: PMC16394
- DOI: 10.1073/pnas.96.8.4692
Stable expression of human beta1,4-galactosyltransferase in plant cells modifies N-linked glycosylation patterns
Abstract
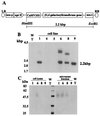
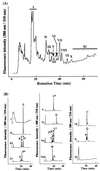
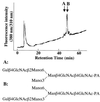
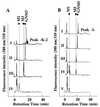
beta1,4-Galactosyltransferase (UDP galactose: beta-N-acetylglucosaminide: beta1,4-galactosyltransferase; EC 2.4.1. 22) catalyzes the transfer of galactose from UDP-Gal to N-acetylglucosamine in the penultimate stages of the terminal glycosylation of N-linked complex oligosaccharides in mammalian cells. Tobacco BY2 cells lack this Golgi enzyme. To determine to what extent the production of a mammalian glycosyltransferase can alter the glycosylation pathway of plant cells, tobacco BY2 suspension-cultured cells were stably transformed with the full-length human galactosyltransferase gene placed under the control of the cauliflower mosaic virus 35S promoter. The expression was confirmed by assaying enzymatic activity as well as by Southern and Western blotting. The transformant with the highest level of enzymatic activity has glycans with galactose residues at the terminal nonreducing ends, indicating the successful modification of the plant cell N-glycosylation pathway. Analysis of the oligosaccharide structures shows that the galactosylated N-glycans account for 47.3% of the total sugar chains. In addition, the absence of the dominant xylosidated- and fucosylated-type sugar chains confirms that the transformed cells can be used to produce glycoproteins without the highly immunogenic glycans typically found in plants. These results demonstrate the synthesis in plants of N-linked glycans with modified and defined sugar chain structures similar to mammalian glycoproteins.
Figures


References
-
- Cramer C L, Weissenborn D L, Oishi K K, Grabau E A, Bennett S, Ponce E, Grabowski G A, Radin D N. Ann N Y Acad Sci. 1996;792:62–71. - PubMed
-
- Kusnadi A R, Nikolov Z L, Howard J A. Biotechnol Bioeng. 1997;56:473–484. - PubMed
-
- Arntzen C J. Nat Biotechnol. 1997;15:221–222. - PubMed
-
- Pen J. In: Transgenic Plants: A Production System for Industrial and Pharmaceutical Proteins. Owen M R L, Pen J, editors. New York: Wiley; 1996. pp. 149–167.
-
- Kukuruzinska M A, Lennon K. Crit Rev Oral Biol Med. 1998;9:415–448. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

