Prospective six year follow up of patients withdrawn from a randomised study comparing parenteral gold salt and methotrexate
- PMID: 10225812
- PMCID: PMC1752873
- DOI: 10.1136/ard.58.5.281
Prospective six year follow up of patients withdrawn from a randomised study comparing parenteral gold salt and methotrexate
Abstract
Objective: To confirm the impression of a better outcome of patients withdrawn from parenteral gold salt therapy compared with those withdrawn from methotrexate.
Methods: Patients with early, active, and erosive RA were randomised for a double blind trial to receive either weekly 15 mg intramuscular methotrexate or 50 mg goldsodiumthiomalate. If the drug had to be withdrawn because of side effects treatment was continued with the other drug in still active disease. Patients with insufficient response were treated with a combination of both drugs. All patients were followed up by an extended clinical and radiographic evaluation.
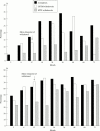
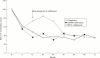
Results: 64 patients each were allocated to methotrexate and gold treatment. After 72 months a complete record was available for 88% of patients. Within the first 36 months 38 patients withdrew from gold treatment (95% because of side effects) and 23 patients withdrew from methotrexate (57% because of side effects). A significant 40% to 70% improvement of all parameters (erythrocyte sedimentation rate, C reactive protein, swollen and tender joints, radiological progression) compared with baseline was observed in patients completing their randomised treatment with gold or methotrexate. The same improvement over three years was seen in patients who withdrew from gold treatment, while patients withdrawing from methotrexate experienced a deterioration of their disease.
Conclusion: Withdrawals represent the majority of patients in long term drug trials. Patients with early RA stopping gold because of side effects show almost the same sustained improvement as patients continuing gold or methotrexate. Patients withdrawn from methotrexate experience a reactivation of their disease.
Figures


Similar articles
-
Comparison of intramuscular methotrexate and gold sodium thiomalate in the treatment of early erosive rheumatoid arthritis: 12 month data of a double-blind parallel study of 174 patients.Br J Rheumatol. 1997 Mar;36(3):345-52. doi: 10.1093/rheumatology/36.3.345. Br J Rheumatol. 1997. PMID: 9133967 Clinical Trial.
-
A 36 month comparative trial of methotrexate and gold sodium thiomalate in the treatment of early active and erosive rheumatoid arthritis.Br J Rheumatol. 1998 Oct;37(10):1060-8. doi: 10.1093/rheumatology/37.10.1060. Br J Rheumatol. 1998. PMID: 9825744 Clinical Trial.
-
Progression in early erosive rheumatoid arthritis: 12 month results from a randomized controlled trial comparing methotrexate and gold sodium thiomalate.Br J Rheumatol. 1998 Nov;37(11):1220-6. doi: 10.1093/rheumatology/37.11.1220. Br J Rheumatol. 1998. PMID: 9851273 Clinical Trial.
-
[Gold preparations in therapy of rheumatoid and psoriatic arthritis].Ter Arkh. 2003;75(5):88-90. Ter Arkh. 2003. PMID: 12847908 Review. Russian. No abstract available.
-
[Does parenteral gold inhibit roentgen progression of chronic polyarthritis?].Z Rheumatol. 1996 Sep-Oct;55(5):307-18. Z Rheumatol. 1996. PMID: 9036717 Review. German.
Cited by
-
Have traditional DMARDs had their day? Effectiveness of parenteral gold compared to biologic agents.Clin Rheumatol. 2005 Jun;24(3):189-202. doi: 10.1007/s10067-004-0869-8. Epub 2004 Jul 24. Clin Rheumatol. 2005. PMID: 15940552 Review.
-
[Methotrexate].Z Rheumatol. 2016 Aug;75(6):599-603. doi: 10.1007/s00393-016-0118-y. Z Rheumatol. 2016. PMID: 27365027 Review. German. No abstract available.
-
Ex vivo effect of gold nanoparticles on porcine synovial membrane.Tissue Barriers. 2013 Apr 1;1(2):e24314. doi: 10.4161/tisb.24314. Tissue Barriers. 2013. PMID: 24665389 Free PMC article.
-
Long-term safety of methotrexate monotherapy in patients with rheumatoid arthritis: a systematic literature research.Ann Rheum Dis. 2009 Jul;68(7):1100-4. doi: 10.1136/ard.2008.093690. Epub 2008 Dec 5. Ann Rheum Dis. 2009. PMID: 19060002 Free PMC article.
-
[Gold as an alternative in the treatment of RA patients with malignancies].Z Rheumatol. 2016 Oct;75(8):834-835. doi: 10.1007/s00393-016-0195-y. Z Rheumatol. 2016. PMID: 27557776 German. No abstract available.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

