Enhancement of B7-1 (CD80) expression on B-lymphoma cells by irradiation
- PMID: 10233753
- PMCID: PMC2326785
- DOI: 10.1046/j.1365-2567.1999.00720.x
Enhancement of B7-1 (CD80) expression on B-lymphoma cells by irradiation
Abstract
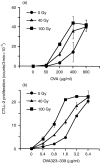
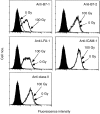
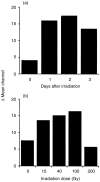
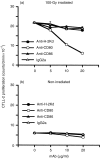
Irradiation of A20.2J mouse B-lymphoma cells enhanced their antigen-presenting ability to induce interleukin-2 (IL-2) production by 42-6A T cells specific for ovalbumin (OVA)323-339/I-Ad. Irradiated and fixed A20.2J cells were more efficient antigen-presenting cells (APC) to present OVA323-339 peptide than the unirradiated and fixed cells. Irradiation selectively increased the expression of B7-1 molecules, but not of the major histocompatibility complex class II molecules, B7-2, lymphocyte function-associated antigen-1, or intracellular adhesion molecule-1. Irradiation of A20.2J cells with 100 Gy followed by overnight incubation was optimal for the enhancement of B7-1 expression. Anti-B7-1 monoclonal antibody inhibited the irradiation-induced enhancement of APC function. Irradiation of A20.2J cells induced the accumulation of B7-1 mRNA. Thus, it was concluded that the enhancement of APC function by irradiation was due to the up-regulation of B7-1 molecules through the accumulation of its mRNA. Although partial inhibition of protein synthesis has been shown to enhance the accumulation of B7-1 mRNA and its expression, irradiation did not decrease the protein synthesis in A20.2J cells. The incubation with irradiated A20.2J cells stimulated unirradiated A20.2J cells to increase B7-1 expression, suggesting that irradiation of A20.2J cells induced expression or secretion of some molecule(s) to enhance B7-1 expression.
Figures

Similar articles
-
Irradiation up-regulates CD80 expression through induction of tumour necrosis factor-alpha and CD40 ligand expression on B lymphoma cells.Immunology. 2002 Jul;106(3):354-62. doi: 10.1046/j.1365-2567.2002.01434.x. Immunology. 2002. PMID: 12100723 Free PMC article.
-
Enhancement of antigen-presenting ability of B lymphoma cells by partial inhibition of protein synthesis through inducing B7-1 expression.Immunology. 1997 Jun;91(2):212-8. doi: 10.1046/j.1365-2567.1997.00253.x. Immunology. 1997. PMID: 9227319 Free PMC article.
-
Irradiation up-regulates CD80 expression through two different mechanisms in spleen B cells, B lymphoma cells, and dendritic cells.Immunology. 2004 Jun;112(2):219-27. doi: 10.1111/j.1365-2567.2004.01872.x. Immunology. 2004. PMID: 15147565 Free PMC article.
-
Simultaneous expression of allogenic class II MHC and B7.1 (CD80) molecules in A20 B-lymphoma cell line enhances tumor immunogenicity.Mol Cells. 2002 Feb 28;13(1):130-6. Mol Cells. 2002. PMID: 11911464
-
From the study of tumor cell immunogenicity to the generation of antitumor cytotoxic cells in non-Hodgkin's lymphomas.Leuk Lymphoma. 2000 Jul;38(3-4):247-63. doi: 10.3109/10428190009087016. Leuk Lymphoma. 2000. PMID: 10830732 Review.
Cited by
-
Disparate effects of depletion of CD1d-reactive T cells during early versus late stages of disease in a genetically susceptible model of lupus.Lupus. 2012 Apr;21(5):485-90. doi: 10.1177/0961203311428459. Epub 2011 Nov 7. Lupus. 2012. PMID: 22065098 Free PMC article.
-
Cell clustering and delay/arrest in T-cell division implicate a novel mechanism of immune modulation by E. coli heat-labile enterotoxin B-subunits.Cell Immunol. 2015 Jun;295(2):150-62. doi: 10.1016/j.cellimm.2015.02.014. Epub 2015 Mar 5. Cell Immunol. 2015. PMID: 25880107 Free PMC article.
-
Rap1 functions as a key regulator of T-cell and antigen-presenting cell interactions and modulates T-cell responses.Mol Cell Biol. 2002 Feb;22(4):1001-15. doi: 10.1128/MCB.22.4.1001-1015.2002. Mol Cell Biol. 2002. PMID: 11809793 Free PMC article.
-
A Century of Radiation Therapy and Adaptive Immunity.Front Immunol. 2017 Apr 11;8:431. doi: 10.3389/fimmu.2017.00431. eCollection 2017. Front Immunol. 2017. PMID: 28443099 Free PMC article. Review.
-
Irradiation up-regulates CD80 expression through induction of tumour necrosis factor-alpha and CD40 ligand expression on B lymphoma cells.Immunology. 2002 Jul;106(3):354-62. doi: 10.1046/j.1365-2567.2002.01434.x. Immunology. 2002. PMID: 12100723 Free PMC article.
References
-
- Mueller DL, Jenkins MK, Schwartz RH. Clonal expansion versus functional clonal inactivation: a costimulatory signaling pathway determines the outcome of T cell antigen receptor occupancy. Annu Rev Immunol. 1989;7:445. - PubMed
-
- Linsley PS, Ledbetter JA. The role of CD28 receptor during T cell responses to antigen. Annu Rev Immunol. 1993;11:191. - PubMed
-
- Galvin F, Freeman GJ, Razi-Wolf Z, et al. Murine B7 antigen provides a sufficient costimulator signal for antigen-specific and MHC-restricted T cell activation. J Immunol. 1992;149:3802. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

