Highly efficient induction of protective immunity by a vaccinia virus vector defective in late gene expression
- PMID: 10233911
- PMCID: PMC112493
- DOI: 10.1128/JVI.73.6.4536-4542.1999
Highly efficient induction of protective immunity by a vaccinia virus vector defective in late gene expression
Abstract
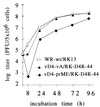
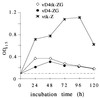
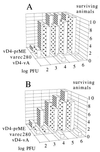
Vaccinia viruses defective in the essential gene coding for the enzyme uracil DNA glycosylase (UDG) do not undergo DNA replication and do not express late genes in wild-type cells. A UDG-deficient vaccinia virus vector carrying the tick-borne encephalitis (TBE) virus prM/E gene, termed vD4-prME, was constructed, and its potential as a vaccine vector was evaluated. High-level expression of the prM/E antigens could be demonstrated in infected complementing cells, and moderate levels were found under noncomplementing conditions. The vD4-prME vector was used to vaccinate mice; animals receiving single vaccination doses as low as 10(4) PFU were fully protected against challenge with high doses of virulent TBE virus. Single vaccination doses of 10(3) PFU were sufficient to induce significant neutralizing antibody titers. With the corresponding replicating virus, doses at least 10-fold higher were needed to achieve protection. The data indicate that late gene expression of the vaccine vector is not required for successful vaccination; early vaccinia virus gene expression induces a potent protective immune response. The new vaccinia virus-based defective vectors are therefore promising live vaccines for prophylaxis and cancer immunotherapy.
Figures
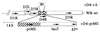

Similar articles
-
Immunogenicity and safety of defective vaccinia virus lister: comparison with modified vaccinia virus Ankara.J Virol. 2002 Aug;76(15):7713-23. doi: 10.1128/jvi.76.15.7713-7723.2002. J Virol. 2002. PMID: 12097585 Free PMC article.
-
Tick-borne Langat/mosquito-borne dengue flavivirus chimera, a candidate live attenuated vaccine for protection against disease caused by members of the tick-borne encephalitis virus complex: evaluation in rhesus monkeys and in mosquitoes.J Virol. 2001 Sep;75(17):8259-67. doi: 10.1128/jvi.75.17.8259-8267.2001. J Virol. 2001. PMID: 11483771 Free PMC article.
-
Stable and Highly Immunogenic MicroRNA-Targeted Single-Dose Live Attenuated Vaccine Candidate against Tick-Borne Encephalitis Constructed Using Genetic Backbone of Langat Virus.mBio. 2019 Apr 23;10(2):e02904-18. doi: 10.1128/mBio.02904-18. mBio. 2019. PMID: 31015334 Free PMC article.
-
Pox viruses as eukaryotic cloning and expression vectors: future medical and veterinary vaccines.Prog Vet Microbiol Immunol. 1988;4:197-217. Prog Vet Microbiol Immunol. 1988. PMID: 3078866 Review.
-
Replicating and host-restricted non-replicating vaccinia virus vectors for vaccine development.Dev Biol Stand. 1994;82:55-63. Dev Biol Stand. 1994. PMID: 7958483 Review.
Cited by
-
A molecular evaluation of dengue virus pathogenesis and its latest vaccine strategies.Mol Biol Rep. 2011 Aug;38(6):3731-40. doi: 10.1007/s11033-010-0488-1. Epub 2010 Nov 24. Mol Biol Rep. 2011. PMID: 21107723 Review.
-
Vectors based on modified vaccinia Ankara expressing influenza H5N1 hemagglutinin induce substantial cross-clade protective immunity.PLoS One. 2011 Jan 24;6(1):e16247. doi: 10.1371/journal.pone.0016247. PLoS One. 2011. PMID: 21283631 Free PMC article.
-
Smallpox vaccines for biodefense.Vaccine. 2009 Nov 5;27 Suppl 4:D73-9. doi: 10.1016/j.vaccine.2009.07.103. Vaccine. 2009. PMID: 19837292 Free PMC article. Review.
-
Expanding the repertoire of Modified Vaccinia Ankara-based vaccine vectors via genetic complementation strategies.PLoS One. 2009;4(5):e5445. doi: 10.1371/journal.pone.0005445. Epub 2009 May 6. PLoS One. 2009. PMID: 19421328 Free PMC article.
-
Development of a novel virus-like particle-based vaccine for preventing tick-borne encephalitis virus infection.Virol Sin. 2023 Oct;38(5):767-777. doi: 10.1016/j.virs.2023.06.003. Epub 2023 Jun 14. Virol Sin. 2023. PMID: 37328107 Free PMC article.
References
-
- Antoine G, Scheiflinger F, Dorner F, Falkner F G. Genomic sequence of the modified vaccinia Ankara (MVA) strain: comparison with other orthopoxviruses. Virology. 1998;244:365–396. - PubMed
-
- Chakrabarti S, Sisler J R, Moss B. Compact, synthetic, vaccinia virus early/late promoter for protein expression. BioTechniques. 1997;23:1094–1097. - PubMed
-
- Coupar B E, Andrew M E, Both G W, Boyle D B. Temporal regulation of influenza hemagglutinin expression in vaccinia virus recombinants and effects on the immune response. Eur J Immunol. 1986;16:1479–1487. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

