Muscarinic control of cytoskeleton in perisynaptic glia
- PMID: 10234016
- PMCID: PMC6782737
- DOI: 10.1523/JNEUROSCI.19-10-03836.1999
Muscarinic control of cytoskeleton in perisynaptic glia
Abstract
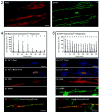
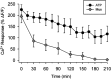
Similar to astrocytes at CNS synapses, perisynaptic Schwann cells (PSCs) surround nerve terminals at the neuromuscular junction (NMJ). These special teloglial cells are sensitive to neurotransmitters and upregulate glial fibrillary acidic protein (GFAP) when deprived of synaptic activity. We found that activation of muscarinic acetylcholine receptors (mAChRs) at PSCs, but not purinergic (ATP and adenosine) or peptidergic [substance P (SP) and calcitonin gene-related peptide (CGRP)] receptors, prevented this upregulation. When applied onto single PSCs, muscarine evoked Ca2+ responses that fatigued but prevented upregulation of this glial cytoskeletal protein. Application of ATP onto single PSCs evoked Ca2+ signals that showed little fatigue, and GFAP upregulation occurred. Thus, Ca2+ signals alone cannot prevent GFAP upregulation in the PSCs. After blockade of cholinergic receptors by gallamine, neuronal activity was not effective in maintaining low GFAP levels in the perisynaptic glia. Last, immunohistochemistry disclosed mAChRs on PSCs and nearby fibroblasts. Thus, acetylcholine secreted by the nerve terminal acts on the PSCs via mAChRs to regulate GFAP. Cytoskeletal changes may influence perisynaptic glial functions, including growth, remodeling, and modulation of the synapse.
Figures



Similar articles
-
Muscarinic Ca2+ responses resistant to muscarinic antagonists at perisynaptic Schwann cells of the frog neuromuscular junction.J Physiol. 1997 Oct 15;504 ( Pt 2)(Pt 2):337-47. doi: 10.1111/j.1469-7793.1997.337be.x. J Physiol. 1997. PMID: 9365908 Free PMC article.
-
Endogenous peptidergic modulation of perisynaptic Schwann cells at the frog neuromuscular junction.J Physiol. 1998 Oct 1;512 ( Pt 1)(Pt 1):197-209. doi: 10.1111/j.1469-7793.1998.197bf.x. J Physiol. 1998. PMID: 9729629 Free PMC article.
-
Synapse-glia interactions at the mammalian neuromuscular junction.J Neurosci. 2001 Jun 1;21(11):3819-29. doi: 10.1523/JNEUROSCI.21-11-03819.2001. J Neurosci. 2001. PMID: 11356870 Free PMC article.
-
Neuron-glia interactions at the neuromuscular synapse.Novartis Found Symp. 2006;276:222-9; discussion 229-37, 275-81. Novartis Found Symp. 2006. PMID: 16805433 Review.
-
Synapse-glia interactions at the vertebrate neuromuscular junction.Neuroscientist. 2005 Oct;11(5):503-13. doi: 10.1177/1073858405277409. Neuroscientist. 2005. PMID: 16151050 Review.
Cited by
-
Structure and Function of the Mammalian Neuromuscular Junction.Compr Physiol. 2022 Aug 11;12(4):3731-3766. doi: 10.1002/cphy.c210022. Compr Physiol. 2022. PMID: 35950651 Free PMC article.
-
Perisynaptic Schwann Cells at the Neuromuscular Synapse: Adaptable, Multitasking Glial Cells.Cold Spring Harb Perspect Biol. 2015 Aug 20;7(10):a020503. doi: 10.1101/cshperspect.a020503. Cold Spring Harb Perspect Biol. 2015. PMID: 26430218 Free PMC article. Review.
-
Properties of Glial Cell at the Neuromuscular Junction Are Incompatible with Synaptic Repair in the SOD1G37R ALS Mouse Model.J Neurosci. 2020 Sep 30;40(40):7759-7777. doi: 10.1523/JNEUROSCI.1748-18.2020. Epub 2020 Aug 28. J Neurosci. 2020. PMID: 32859714 Free PMC article.
-
Excitatory neurotransmission activates compartmentalized calcium transients in Müller glia without affecting lateral process motility.Elife. 2021 Dec 16;10:e73202. doi: 10.7554/eLife.73202. Elife. 2021. PMID: 34913435 Free PMC article.
-
Glial processes at the Drosophila larval neuromuscular junction match synaptic growth.PLoS One. 2012;7(5):e37876. doi: 10.1371/journal.pone.0037876. Epub 2012 May 29. PLoS One. 2012. PMID: 22666403 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
