Developing Schwann cells acquire the ability to survive without axons by establishing an autocrine circuit involving insulin-like growth factor, neurotrophin-3, and platelet-derived growth factor-BB
- PMID: 10234017
- PMCID: PMC6782711
- DOI: 10.1523/JNEUROSCI.19-10-03847.1999
Developing Schwann cells acquire the ability to survive without axons by establishing an autocrine circuit involving insulin-like growth factor, neurotrophin-3, and platelet-derived growth factor-BB
Abstract
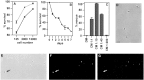
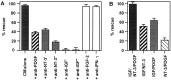
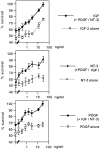
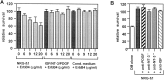
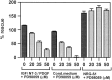
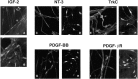
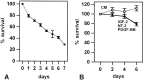
Although Schwann cell precursors from early embryonic nerves die in the absence of axonal signals, Schwann cells in older nerves can survive in the absence of axons in the distal stump of transected nerves. This is crucially important, because successful axonal regrowth in a damaged nerve depends on interactions with living Schwann cells in the denervated distal stump. Here we show that Schwann cells acquire the ability to survive without axons by establishing an autocrine survival loop. This mechanism is absent in precursors. We show that insulin-like growth factor, neurotrophin-3, and platelet-derived growth factor-BB are important components of this autocrine survival signal. The secretion of these factors by Schwann cells has significant implications for cellular communication in developing nerves, in view of their known ability to regulate survival and differentiation of other cells including neurons.
Figures



References
-
- Alessi DR, Cuenda A, Cohen P, Dudley DT, Saltiel AR. PD 098059 is a specific inhibitor of the activation of mitogen-activated protein kinase kinase in vitro and in vivo. J Biol Chem. 1995;270:27489–27494. - PubMed
-
- Barres BA, Hart IK, Coles HSR, Burne JF, Voyvodic JT, Richardson WD, Raff MC. Cell death and control of cell survival in the oligodendrocyte lineage. Cell. 1992;70:31–46. - PubMed
-
- Barres BA, Jacobson MD, Schmid R, Sendtner M, Raff MC. Does oligodendrocyte survival depend on axons? Curr Biol. 1993a;3:489–497. - PubMed
-
- Barres BA, Schmid R, Sendtner M, Raff MC. Multiple extracellular signals are required for long-term oligodendrocyte survival. Development. 1993b;118:283–295. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
