Specific, rapid synthesis of Phe-RNA by RNA
- PMID: 10318907
- PMCID: PMC21883
- DOI: 10.1073/pnas.96.10.5470
Specific, rapid synthesis of Phe-RNA by RNA
Abstract
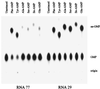
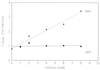
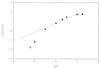
RNA 77, derived by selection amplification, accelerates its own conversion to Phe-RNA (relative to randomized RNA) more than 6 x 10(7)-fold, by using amino acid adenylates as substrate. A modified assay system allows measurement of slow rates of aa-RNA formation, which for disfavored amino acid substrates can be more than 10(4)-fold slower than phenylalanine. Thus unlike previously characterized self-aminoacylators, RNA 77 catalysis is highly amino acid selective. Remarkably, both rates of aminoacyl transfer and amino acid specificities are greater for RNA 77 than measured for protein PheRS. These data experimentally support the possible existence of an ancestral amino acid-specific translation system relying entirely on RNA catalysis. RNA 77 itself embodies a possible transitional evolutionary state, in which side-chain-specific aa-RNA formation and anticodon-codon pairing were invested in the same molecule.
Figures




Similar articles
-
Aminoacyl-RNA synthesis catalyzed by an RNA.Science. 1995 Feb 3;267(5198):643-7. doi: 10.1126/science.7530860. Science. 1995. PMID: 7530860
-
A tiny RNA that catalyzes both aminoacyl-RNA and peptidyl-RNA synthesis.RNA. 1999 Nov;5(11):1482-9. doi: 10.1017/s1355838299991264. RNA. 1999. PMID: 10580476 Free PMC article.
-
Small-molecule-substrate interactions with a self-aminoacylating ribozyme.J Mol Biol. 1997 May 9;268(3):631-9. doi: 10.1006/jmbi.1997.0988. J Mol Biol. 1997. PMID: 9171286
-
Genomics and the evolution of aminoacyl-tRNA synthesis.Acta Biochim Pol. 2001;48(2):313-21. Acta Biochim Pol. 2001. PMID: 11732603 Review.
-
Three-dimensional structure of transfer RNA and its functional implications.Adv Enzymol Relat Areas Mol Biol. 1978;46:279-315. doi: 10.1002/9780470122914.ch4. Adv Enzymol Relat Areas Mol Biol. 1978. PMID: 205095 Review. No abstract available.
Cited by
-
The scene of a frozen accident.RNA. 2000 Apr;6(4):485-98. doi: 10.1017/s1355838200000224. RNA. 2000. PMID: 10786840 Free PMC article.
-
Arginine cofactors on the polymerase ribozyme.PLoS One. 2011;6(9):e25030. doi: 10.1371/journal.pone.0025030. Epub 2011 Sep 20. PLoS One. 2011. PMID: 21949841 Free PMC article.
-
Rapid and simple ribozymic aminoacylation using three conserved nucleotides.J Am Chem Soc. 2009 Apr 15;131(14):5257-63. doi: 10.1021/ja809419f. J Am Chem Soc. 2009. PMID: 19351205 Free PMC article.
-
Triplet-Encoded Prebiotic RNA Aminoacylation.J Am Chem Soc. 2023 Jul 26;145(29):15971-15980. doi: 10.1021/jacs.3c03931. Epub 2023 Jul 12. J Am Chem Soc. 2023. PMID: 37435826 Free PMC article.
-
The Origin of Translation: Bridging the Nucleotides and Peptides.Int J Mol Sci. 2022 Dec 22;24(1):197. doi: 10.3390/ijms24010197. Int J Mol Sci. 2022. PMID: 36613641 Free PMC article. Review.
References
-
- Illangasekare M, Sanchez G, Nickles T, Yarus M. Science. 1995;267:643–647. - PubMed
-
- Piccirilli J A, McConnell T S, Zaug A J, Noller H F, Cech T R. Science. 1992;256:1420–1424. - PubMed
-
- Dai X-C, de Mesmaeker A, Joyce G F. Science. 1995;267:237–240. - PubMed
-
- Joyce G F, Dai X-C, De Mesmaeker A. Science. 1996;272:18–19. - PubMed
-
- Lohse P A, Szostak J W. Nature (London) 1996;381:442–444. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

