Expression and functional characteristics of calpain 3 isoforms generated through tissue-specific transcriptional and posttranscriptional events
- PMID: 10330145
- PMCID: PMC104364
- DOI: 10.1128/MCB.19.6.4047
Expression and functional characteristics of calpain 3 isoforms generated through tissue-specific transcriptional and posttranscriptional events
Abstract
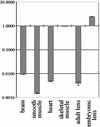
Calpain 3 is a nonlysosomal cysteine protease whose biological functions remain unknown. We previously demonstrated that this protease is altered in limb girdle muscular dystrophy type 2A patients. Preliminary observations suggested that its gene is subjected to alternative splicing. In this paper, we characterize transcriptional and posttranscriptional events leading to alterations involving the NS, IS1, and IS2 regions and/or the calcium binding domains of the mouse calpain 3 gene (capn3). These events can be divided into three groups: (i) splicing of exons that preserve the translation frame, (ii) inclusion of two distinct intronic sequences between exons 16 and 17 that disrupt the frame and would lead, if translated, to a truncated protein lacking domain IV, and (iii) use of an alternative first exon specific to lens tissue. In addition, expression of these isoforms seems to be regulated. Investigation of the proteolytic activities and titin binding abilities of the translation products of some of these isoforms clearly indicated that removal of these different protein segments affects differentially the biochemical properties examined. In particular, removal of exon 6 impaired the autolytic but not fodrinolytic activity and loss of exon 16 led to an increased titin binding and a loss of fodrinolytic activity. These results are likely to impact our understanding of the pathophysiology of calpainopathies and the development of therapeutic strategies.
Figures





References
-
- Bucher P. Weight matrix descriptions of four eukaryotic RNA polymerase II promoter elements derived from 502 unrelated promoter sequences. J Mol Biol. 1989;212:563–578. - PubMed
-
- David L L, Shearer T R. Purification of calpain II from rat lens and determination of endogenous substrates. Exp Eye Res. 1986;42:227–238. - PubMed
-
- Fardeau M, Hillaire D, Mignard C, Feingold N, Mignard D, de Ubeda B, Collin H, Tomé F M S, Richard I, Beckmann J S. Juvenile limb-girdle muscular dystrophy: clinical, histopathological, and genetic data from a small community living in the Reunion Island. Brain. 1996;119:295–308. - PubMed
-
- Fougerousse F, Durand M, Suel L, Pourquié O, Delezoide A-L, Roméro N, Abitbol M, Beckmann J S. Expression of genes (CAPN3, SGCA, SGCB and TTN) involved in progressive muscular dystrophies during early human development. Genomics. 1998;48:145–156. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
