Estrogen's bone-protective effects may involve differential IL-1 receptor regulation in human osteoclast-like cells
- PMID: 10330423
- PMCID: PMC408450
- DOI: 10.1172/JCI4682
Estrogen's bone-protective effects may involve differential IL-1 receptor regulation in human osteoclast-like cells
Abstract
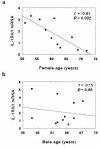
Declining estrogen levels during the first postmenopausal decade lead to rapid bone loss and increased fracture risk that can be reversed by estrogen replacement therapy. The bone-protective effects of estrogen may involve suppression of inflammatory cytokines that promote osteoclastogenesis and bone resorption, such as IL-1, TNF-alpha, and IL-6. We investigated whether estrogen modulates IL-1 actions on human osteoclasts (OCs) and other bone cell types. Isolated human OCs and primary bone marrow-derived OC-like cells expressed both the signaling (IL-1RI) and decoy (IL-1RII) IL-1 receptors, whereas only IL-1RI was detected in osteoblasts. IL-1RII/IL-1RI mRNA ratios and release of soluble IL-1RII (sIL-1RII) were lower in OC-like cells derived from women in the late postmenopausal period compared with younger women, but were unrelated to male donor age, suggesting that estrogen might play a role in regulating IL-1 receptor levels in vivo. Estrogen directly reduced in vitro OC-like cell IL-1RI mRNA levels while increasing IL-1RII mRNA levels and sIL-1RII release. These estrogenic events were associated with inhibited IL-1-mediated cytokine (IL-8) mRNA induction and cell survival, i.e., increased apoptosis. In contrast, estrogen did not alter IL-1R levels or IL-1 responsiveness in primary human osteoblasts or bone marrow stromal cells. We conclude that one novel mechanism by which estrogen exerts bone-protective effects may include a selective modulation of IL-1R isoform levels in OC or OC-like cells, thereby reducing their IL-1 responsiveness and cell survival. Conversely, this restraint on IL-1 actions may be lost as estrogen levels decline in aging women, contributing to an enhanced OC-mediated postmenopausal bone loss.
Figures


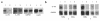
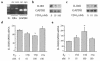
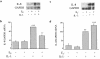
Similar articles
-
Cellular and molecular effects of growth hormone and estrogen on human bone cells.APMIS Suppl. 1997;71:1-30. APMIS Suppl. 1997. PMID: 9357492 Review.
-
Cytokine RNA levels in transiliac bone biopsies from healthy early postmenopausal women.Bone. 2000 Feb;26(2):137-45. doi: 10.1016/s8756-3282(99)00260-4. Bone. 2000. PMID: 10678408 Clinical Trial.
-
The type II IL-1 receptor interacts with the IL-1 receptor accessory protein: a novel mechanism of regulation of IL-1 responsiveness.J Immunol. 1998 Dec 15;161(12):6871-7. J Immunol. 1998. PMID: 9862719
-
Interleukin-1 receptors type I and type II are differentially regulated in human keratinocytes by ultraviolet B radiation.J Invest Dermatol. 1996 Dec;107(6):865-70. J Invest Dermatol. 1996. PMID: 8941676
-
Estrogen, cytokines, and pathogenesis of postmenopausal osteoporosis.J Bone Miner Res. 1996 Aug;11(8):1043-51. doi: 10.1002/jbmr.5650110802. J Bone Miner Res. 1996. PMID: 8854239 Review.
Cited by
-
Sex-related survival differences in murine cardiomyopathy are associated with differences in TNF-receptor expression.J Clin Invest. 2000 Aug;106(4):589-97. doi: 10.1172/JCI9307. J Clin Invest. 2000. PMID: 10953034 Free PMC article.
-
Molecular mechanisms and clinical management of cancer bone metastasis.Bone Res. 2020 Jul 29;8(1):30. doi: 10.1038/s41413-020-00105-1. eCollection 2020. Bone Res. 2020. PMID: 32793401 Free PMC article. Review.
-
Gender differences in neurological disease: role of estrogens and cytokines.Endocrine. 2006 Apr;29(2):243-56. doi: 10.1385/ENDO:29:2:243. Endocrine. 2006. PMID: 16785600 Review.
-
Absence of platelet-activating factor receptor protects mice from osteoporosis following ovariectomy.J Clin Invest. 2004 Jul;114(1):85-93. doi: 10.1172/JCI20504. J Clin Invest. 2004. PMID: 15232615 Free PMC article.
-
Analyses of the association between breast cancer and osteoporosis/fracture history: a cross-sectional study using KoGES HEXA data.Arch Osteoporos. 2021 Jun 19;16(1):98. doi: 10.1007/s11657-021-00947-0. Arch Osteoporos. 2021. PMID: 34148148
References
-
- Turner R, Riggs B, Spelsberg T. Skeletal effects of estrogen. Endocr Rev. 1994;15:275–300. - PubMed
-
- Smith E, et al. Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man. N Engl J Med. 1994;331:1056–1061. - PubMed
-
- Korach K, et al. Estrogen receptor gene disruption: molecular characterization and experimental and clinical phenotypes. Recent Prog Horm Res. 1996;51:159–186. - PubMed
-
- Horowitz M. Cytokines and estrogen in bone: anti-osteoporotic effects. Science. 1993;260:626–627. - PubMed
-
- Pensler JM, Radosevich JA, Higbee R, Langman CB. Osteoclasts isolated from membranous bone in children exhibit nuclear estrogen and progesterone receptors. J Bone Miner Res. 1990;5:797–802. - PubMed

