Modulation of major histocompatibility complex protein expression by human gamma interferon mediated by cysteine proteinase-adhesin polyproteins of Porphyromonas gingivalis
- PMID: 10338509
- PMCID: PMC96610
- DOI: 10.1128/IAI.67.6.2986-2995.1999
Modulation of major histocompatibility complex protein expression by human gamma interferon mediated by cysteine proteinase-adhesin polyproteins of Porphyromonas gingivalis
Abstract
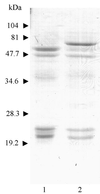
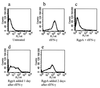
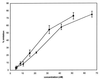
Cysteine proteinases have been emphasized in the virulence of Porphyromonas gingivalis in chronic periodontitis. These hydrolases may promote the degradation of extracellular matrix proteins and disrupt components of the immune system. In this study it was shown that purified Arg-gingipain and Lys-gingipain inhibited expression of class II major histocompatibility complex (MHC) proteins in response to the stimulation of endothelial cells with human gamma interferon (IFN-gamma). Treatment with the cysteine proteinases resulted in a rapid shift in the apparent molecular size of IFN-gamma from 17 to 15 kDa, as shown by Western blot analysis, a response which also occurred in the presence of serum. Further, glycosylated natural IFN-gamma from human leukocytes and unglycosylated recombinant IFN-gamma from Escherichia coli were both digested by the cysteine proteinases. Immunoblot analysis indicated that cleavage within the carboxyl terminus of recombinant IFN-gamma correlated with the loss of induction of MHC class II expression as monitored by analytical flow cytometry. No hydrolysis of MHC class II molecules or human IFN-gamma receptor by these proteinases was detected by Western blot analysis. These findings suggest that P. gingivalis cysteine proteinases may alter the cytokine network at the point of infection through the cleavage of IFN-gamma. Degradation of IFN-gamma could have important consequences for the recruitment and activation of leukocytes and therefore may contribute significantly to the destruction of the periodontal attachment.
Figures
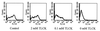





Similar articles
-
The K1K2 region of Lys-gingipain of Porphyromonas gingivalis blocks induction of HLA expression by gamma interferon.Infect Immun. 2012 Oct;80(10):3733-41. doi: 10.1128/IAI.00528-12. Epub 2012 Jul 16. Infect Immun. 2012. PMID: 22802347 Free PMC article.
-
Modulation of gamma interferon-induced major histocompatibility complex class II gene expression by Porphyromonas gingivalis membrane vesicles.Infect Immun. 2002 Mar;70(3):1185-92. doi: 10.1128/IAI.70.3.1185-1192.2002. Infect Immun. 2002. PMID: 11854199 Free PMC article.
-
Cleavage of the human C5A receptor by proteinases derived from Porphyromonas gingivalis: cleavage of leukocyte C5a receptor.Adv Exp Med Biol. 1996;389:155-64. doi: 10.1007/978-1-4613-0335-0_19. Adv Exp Med Biol. 1996. PMID: 8861006
-
Porphyromonas gingivalis gingipains: the molecular teeth of a microbial vampire.Curr Protein Pept Sci. 2003 Dec;4(6):409-26. doi: 10.2174/1389203033487009. Curr Protein Pept Sci. 2003. PMID: 14683427 Review.
-
Suppression of virulence of Porphyromonas gingivalis by potent inhibitors specific for gingipains.Curr Protein Pept Sci. 2003 Dec;4(6):451-8. doi: 10.2174/1389203033486992. Curr Protein Pept Sci. 2003. PMID: 14683430 Review.
Cited by
-
The RgpA-Kgp proteinase-adhesin complexes of Porphyromonas gingivalis Inactivate the Th2 cytokines interleukin-4 and interleukin-5.Infect Immun. 2009 Apr;77(4):1451-8. doi: 10.1128/IAI.01377-08. Epub 2009 Jan 21. Infect Immun. 2009. PMID: 19168731 Free PMC article.
-
Porphyromonas gingivalis gingipain-R enhances interleukin-8 but decreases gamma interferon-inducible protein 10 production by human gingival fibroblasts in response to T-cell contact.Infect Immun. 2001 Jul;69(7):4493-501. doi: 10.1128/IAI.69.7.4493-4501.2001. Infect Immun. 2001. PMID: 11401991 Free PMC article.
-
Functional implication of the hydrolysis of platelet endothelial cell adhesion molecule 1 (CD31) by gingipains of Porphyromonas gingivalis for the pathology of periodontal disease.Infect Immun. 2005 Mar;73(3):1386-98. doi: 10.1128/IAI.73.3.1386-1398.2005. Infect Immun. 2005. PMID: 15731036 Free PMC article.
-
Dichotomy of gingipains action as virulence factors: from cleaving substrates with the precision of a surgeon's knife to a meat chopper-like brutal degradation of proteins.Periodontol 2000. 2010 Oct;54(1):15-44. doi: 10.1111/j.1600-0757.2010.00377.x. Periodontol 2000. 2010. PMID: 20712631 Free PMC article. Review. No abstract available.
-
Emerging family of proline-specific peptidases of Porphyromonas gingivalis: purification and characterization of serine dipeptidyl peptidase, a structural and functional homologue of mammalian prolyl dipeptidyl peptidase IV.Infect Immun. 2000 Mar;68(3):1176-82. doi: 10.1128/IAI.68.3.1176-1182.2000. Infect Immun. 2000. PMID: 10678923 Free PMC article.
References
-
- Arakawa T, Hsu Y R, Parker C G, Lai P H. Role of polycationic C-terminal portion in the structure and activity of recombinant human interferon-gamma. J Biol Chem. 1986;261:8334–8339. - PubMed
-
- Benacerraf B. Role of MHC gene products in immune regulation. Science. 1981;212:1229–1238. - PubMed
-
- Boehm U, Klamp T, Groot M, Howard J C. Cellular responses to interferon-γ. Annu Rev Immunol. 1997;15:749–795. - PubMed
-
- Calkins C C, Platt K, Potempa J, Travis J. Inactivation of tumor necrosis factor-α by proteinases (gingipains) from the periodontal pathogen, Porphyromonas gingivalis: implications of immune evasion. J Biol Chem. 1998;273:6611–6614. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

