Metalloprotease-mediated ligand release regulates autocrine signaling through the epidermal growth factor receptor
- PMID: 10339571
- PMCID: PMC26865
- DOI: 10.1073/pnas.96.11.6235
Metalloprotease-mediated ligand release regulates autocrine signaling through the epidermal growth factor receptor
Abstract
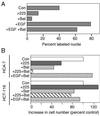
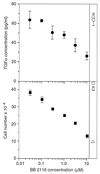
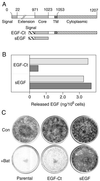
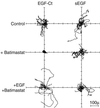
Ligands that activate the epidermal growth factor receptor (EGFR) are synthesized as membrane-anchored precursors that appear to be proteolytically released by members of the ADAM family of metalloproteases. Because membrane-anchored EGFR ligands are thought to be biologically active, the role of ligand release in the regulation of EGFR signaling is unclear. To investigate this question, we used metalloprotease inhibitors to block EGFR ligand release from human mammary epithelial cells. These cells express both transforming growth factor alpha and amphiregulin and require autocrine signaling through the EGFR for proliferation and migration. We found that metalloprotease inhibitors reduced cell proliferation in direct proportion to their effect on transforming growth factor alpha release. Metalloprotease inhibitors also reduced growth of EGF-responsive tumorigenic cell lines and were synergistic with the inhibitory effects of antagonistic EGFR antibodies. Blocking release of EGFR ligands also strongly inhibited autocrine activation of the EGFR and reduced both the rate and persistence of cell migration. The effects of metalloprotease inhibitors could be reversed by either adding exogenous EGF or by expressing an artificial gene for EGF that lacked a membrane-anchoring domain. Our results indicate that soluble rather than membrane-anchored forms of the ligands mediate most of the biological effects of EGFR ligands. Metalloprotease inhibitors have shown promise in preventing spread of metastatic disease. Many of their antimetastatic effects could be the result of their ability to inhibit autocrine signaling through the EGFR.
Figures

References
-
- Miettinen P J, Berger J E, Meneses J, Phung Y, Pedersen R A, Werb Z, Derynck R. Nature (London) 1995;376:337–341. - PubMed
-
- Threadgill D W, Dlugosz A A, Hansen L A, Tennenbaum T, Lichti U, Yee D, LaMantia C, Mourton T, Herrup K, Harris R C, et al. Science. 1995;269:230–234. - PubMed
-
- Derynck R. Adv Cancer Res. 1992;58:27–52. - PubMed
-
- Shoyab M, Plowman G D, McDonald V L, Bradley J G, Todaro G J. Science. 1989;243:1074–1076. - PubMed
-
- Higashiyama S, Abraham J A, Miller J, Fiddes J C, Klagsbrun M. Science. 1991;251:936–939. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

