beta-Trcp couples beta-catenin phosphorylation-degradation and regulates Xenopus axis formation
- PMID: 10339577
- PMCID: PMC26871
- DOI: 10.1073/pnas.96.11.6273
beta-Trcp couples beta-catenin phosphorylation-degradation and regulates Xenopus axis formation
Abstract
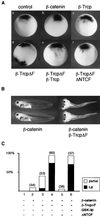
Regulation of beta-catenin stability is essential for Wnt signal transduction during development and tumorigenesis. It is well known that serine-phosphorylation of beta-catenin by the Axin-glycogen synthase kinase (GSK)-3beta complex targets beta-catenin for ubiquitination-degradation, and mutations at critical phosphoserine residues stabilize beta-catenin and cause human cancers. How beta-catenin phosphorylation results in its degradation is undefined. Here we show that phosphorylated beta-catenin is specifically recognized by beta-Trcp, an F-box/WD40-repeat protein that also associates with Skp1, an essential component of the ubiquitination apparatus. beta-catenin harboring mutations at the critical phosphoserine residues escapes recognition by beta-Trcp, thus providing a molecular explanation for why these mutations cause beta-catenin accumulation that leads to cancer. Inhibition of endogenous beta-Trcp function by a dominant negative mutant stabilizes beta-catenin, activates Wnt/beta-catenin signaling, and induces axis formation in Xenopus embryos. Therefore, beta-Trcp plays a central role in recruiting phosphorylated beta-catenin for degradation and in dorsoventral patterning of the Xenopus embryo.
Figures





References
-
- Cadigan K M, Nusse R. Genes Dev. 1997;11:3286–3305. - PubMed
-
- Moon R T, Brown J D, Torres M. Trends Genet. 1997;13:157–162. - PubMed
-
- Kinzler K W, Vogelstein B. Cell. 1996;87:159–170. - PubMed
-
- Peifer M. Science. 1997;275:1752–1754. - PubMed
-
- Behrens J, Von Kries J P, Kuhl M, Bruhn L, Wedlich D, Grosschedl R, Birchmeier W. Nature (London) 1996;382:638–642. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

