GTP bound to chloroplast thylakoid membranes is required for light-induced, multienzyme degradation of the photosystem II D1 protein
- PMID: 10339625
- PMCID: PMC26919
- DOI: 10.1073/pnas.96.11.6547
GTP bound to chloroplast thylakoid membranes is required for light-induced, multienzyme degradation of the photosystem II D1 protein
Abstract
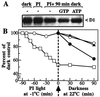
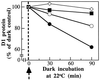
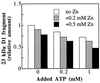
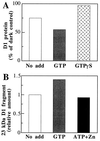
Even though light is the driving force in photosynthesis, it also can be harmful to plants. The water-splitting photosystem II is the main target for this light stress, leading to inactivation of photosynthetic electron transport and photooxidative damage to its reaction center. The plant survives through an intricate repair mechanism involving proteolytic degradation and replacement of the photodamaged reaction center D1 protein. Based on experiments with isolated chloroplast thylakoid membranes and photosystem II core complexes, we report several aspects concerning the rapid turnover of the D1 protein. (i) The primary cleavage step is a GTP-dependent process, leading to accumulation of a 23-kDa N-terminal fragment. (ii) Proteolysis of the D1 protein is inhibited below basal levels by nonhydrolyzable GTP analogues and apyrase treatment, indicating the existence of endogenous GTP tightly bound to the thylakoid membrane. This possibility was corroborated by binding studies. (iii) The proteolysis of the 23-kDa primary degradation fragment (but not of the D1 protein) is an ATP- and zinc-dependent process. (iv) D1 protein degradation is a multienzyme event involving a strategic (primary) protease and a cleaning-up (secondary) protease. (v) The chloroplast FtsH protease is likely to be involved in the secondary degradation steps. Apart from its significance for understanding the repair of photoinhibition, the discovery of tightly bound GTP should have general implications for other regulatory reactions and signal transduction pathways associated with the photosynthetic membrane.
Figures


Similar articles
-
The thylakoid FtsH protease plays a role in the light-induced turnover of the photosystem II D1 protein.Plant Cell. 2000 Mar;12(3):419-31. doi: 10.1105/tpc.12.3.419. Plant Cell. 2000. PMID: 10715327 Free PMC article.
-
FtsH Protease in the Thylakoid Membrane: Physiological Functions and the Regulation of Protease Activity.Front Plant Sci. 2018 Jun 20;9:855. doi: 10.3389/fpls.2018.00855. eCollection 2018. Front Plant Sci. 2018. PMID: 29973948 Free PMC article. Review.
-
Quality control of photosystem II.Plant Cell Physiol. 2001 Feb;42(2):121-8. doi: 10.1093/pcp/pce022. Plant Cell Physiol. 2001. PMID: 11230565 Review.
-
Quality control of Photosystem II: the molecular basis for the action of FtsH protease and the dynamics of the thylakoid membranes.J Photochem Photobiol B. 2014 Aug;137:100-6. doi: 10.1016/j.jphotobiol.2014.02.012. Epub 2014 Mar 4. J Photochem Photobiol B. 2014. PMID: 24725639 Review.
-
A chloroplast DegP2 protease performs the primary cleavage of the photodamaged D1 protein in plant photosystem II.EMBO J. 2001 Feb 15;20(4):713-22. doi: 10.1093/emboj/20.4.713. EMBO J. 2001. PMID: 11179216 Free PMC article.
Cited by
-
Recent Advances in Understanding the Structural and Functional Evolution of FtsH Proteases.Front Plant Sci. 2022 Apr 6;13:837528. doi: 10.3389/fpls.2022.837528. eCollection 2022. Front Plant Sci. 2022. PMID: 35463435 Free PMC article. Review.
-
Clues to the functions of plant NDPK isoforms.Naunyn Schmiedebergs Arch Pharmacol. 2015 Feb;388(2):119-32. doi: 10.1007/s00210-014-1009-x. Epub 2014 Jun 26. Naunyn Schmiedebergs Arch Pharmacol. 2015. PMID: 24964975 Review.
-
Multiple evidence for nucleotide metabolism in the chloroplast thylakoid lumen.Proc Natl Acad Sci U S A. 2004 Feb 3;101(5):1409-14. doi: 10.1073/pnas.0308164100. Epub 2004 Jan 21. Proc Natl Acad Sci U S A. 2004. PMID: 14736920 Free PMC article.
-
Expression, assembly and auxiliary functions of photosystem II oxygen-evolving proteins in higher plants.Photosynth Res. 2007 Jul-Sep;93(1-3):89-100. doi: 10.1007/s11120-007-9154-4. Epub 2007 Mar 23. Photosynth Res. 2007. PMID: 17380423 Review.
-
Structural and functional aspects of the MSP (PsbO) and study of its differences in thermophilic versus mesophilic organisms.Photosynth Res. 2008 Oct-Dec;98(1-3):365-89. doi: 10.1007/s11120-008-9353-7. Epub 2008 Sep 9. Photosynth Res. 2008. PMID: 18780158 Review.
References
-
- Barber J, Andersson B. Trends Biochem Sci. 1992;17:61–66. - PubMed
-
- Prasil O, Adir N, Ohad I. In: Topics in Photosynthesis. Barber J, editor. Vol. 11. Amsterdam: Elsevier; 1992. pp. 295–348.
-
- Andersson B, Salter A H, Virgin I, Vass I, Styring S. J Photochem Photobiol B. 1992;15:15–31.
-
- Hankamer B, Barber J, Boekema E J. Annu Rev Plant Physiol Mol Biol. 1997;48:641–671. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

