Lysylhydroxylation and non-reducible crosslinking of human supraspinatus tendon collagen: changes with age and in chronic rotator cuff tendinitis
- PMID: 10343538
- PMCID: PMC1752756
- DOI: 10.1136/ard.58.1.35
Lysylhydroxylation and non-reducible crosslinking of human supraspinatus tendon collagen: changes with age and in chronic rotator cuff tendinitis
Abstract
Objectives: To investigate age related and site specific variations in turnover and chemistry of the collagen network in healthy tendons as well as the role of collagen remodelling in the degeneration of the supraspinatus tendon (ST-D) in rotator cuff tendinitis.
Methods: Collagen content and the amount of hydroxylysine (Hyl), hydroxy-lysylpyridinoline (HP), lysylpyridinoline (LP), and the degree of non-enzymatic glycation (pentosidine) were investigated in ST-D and in normal human supraspinatus (ST-N) and biceps brachii tendons (BT-N) by high-performance liquid chromatography.
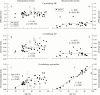
Results: In BT-N, tendons that served as control tissue as it shows rarely matrix abnormalities, pentosidine levels rise linearly with age (20-90 years), indicating little tissue remodelling (resulting in an undisturbed accumulation of pentosidine). A similar accumulation was observed in ST-N up to 50 years. At older ages, little pentosidine accumulation was observed and pentosidine levels showed large interindividual variability. This was interpreted as remodelling of collagen in normal ST after age 50 years because of microruptures (thus diluting old collagen with newly synthesised collagen). All degenerate ST samples showed decreased pentosidine levels compared with age matched controls, indicating extensive remodelling in an attempt to repair the tendon defect. Collagen content and the amount of Hyl, HP, and LP of ST-N and BT-N did not change with age. With the exception of collagen content, which did not differ, all parameters were significantly (p < 0.001) lower in BT-N. The ST-D samples had a reduced collagen content and had higher Hyl, HP, and LP levels than ST-N (p < 0.001).
Conclusions: Inasmuch as Hyl, HP, and LP levels in ST-N did not change with age, tissue remodelling as a consequence of microruptures does not seem to affect the quality of the tendon collagen. On the other hand, the clearly different profile of post-translational modifications in ST-D indicates that the newly deposited collagen network in degenerated tendons is qualitatively different. It is concluded that in ST-D the previously functional and carefully constructed matrix is replaced by aberrant collagen. This may result in a mechanically less stable tendon; as the supraspinatus is constantly subjected to considerable forces this could explain why tendinitis is mostly of a chronic nature.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

