Mapping an interface of SecY (PrlA) and SecE (PrlG) by using synthetic phenotypes and in vivo cross-linking
- PMID: 10348856
- PMCID: PMC93811
- DOI: 10.1128/JB.181.11.3438-3444.1999
Mapping an interface of SecY (PrlA) and SecE (PrlG) by using synthetic phenotypes and in vivo cross-linking
Abstract
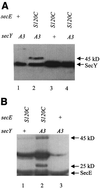
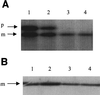
SecY and SecE are integral cytoplasmic membrane proteins that form an essential part of the protein translocation machinery in Escherichia coli. Sites of direct contact between these two proteins have been suggested by the allele-specific synthetic phenotypes exhibited by pairwise combinations of prlA and prlG signal sequence suppressor mutations in these genes. We have introduced cysteine residues within the first periplasmic loop of SecY and the second periplasmic loop of SecE, at a specific pair of positions identified by this genetic interaction. The expression of the cysteine mutant pair results in a dominant lethal phenotype that requires the presence of DsbA, which catalyzes the formation of disulfide bonds. A reducible SecY-SecE complex is also observed, demonstrating that these amino acids must be sufficiently proximal to form a disulfide bond. The use of cysteine-scanning mutagenesis enabled a second contact site to be discovered. Together, these two points of contact allow the modeling of a limited region of quaternary structure, establishing the first characterized site of interaction between these two proteins. This study proves that actual points of protein-protein contact can be identified by using synthetic phenotypes.
Figures
References
-
- Bardwell J C A, McGovern K, Beckwith J. Identification of a protein required for disulfide bond formation in vivo. Cell. 1991;67:581–589. - PubMed
-
- Bieker K L, Phillips G J, Silhavy T J. The sec and prl genes of Escherichia coli. J Bioenerg Biomembr. 1990;22:291–310. - PubMed
-
- Bieker K L, Silhavy T J. PrlA (SecY) and PrlG (SecE) interact directly and function sequentially during protein translocation in E. coli. Cell. 1990;61:833–842. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

