Bioenergetic aspects of halophilism
- PMID: 10357854
- PMCID: PMC98969
- DOI: 10.1128/MMBR.63.2.334-348.1999
Bioenergetic aspects of halophilism
Abstract
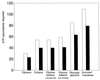
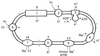
Examination of microbial diversity in environments of increasing salt concentrations indicates that certain types of dissimilatory metabolism do not occur at the highest salinities. Examples are methanogenesis for H2 + CO2 or from acetate, dissimilatory sulfate reduction with oxidation of acetate, and autotrophic nitrification. Occurrence of the different metabolic types is correlated with the free-energy change associated with the dissimilatory reactions. Life at high salt concentrations is energetically expensive. Most bacteria and also the methanogenic Archaea produce high intracellular concentrations of organic osmotic solutes at a high energetic cost. All halophilic microorganisms expend large amounts of energy to maintain steep gradients of NA+ and K+ concentrations across their cytoplasmic membrane. The energetic cost of salt adaptation probably dictates what types of metabolism can support life at the highest salt concentrations. Use of KCl as an intracellular solute, while requiring far-reaching adaptations of the intracellular machinery, is energetically more favorable than production of organic-compatible solutes. This may explain why the anaerobic halophilic fermentative bacteria (order Haloanaerobiales) use this strategy and also why halophilic homoacetogenic bacteria that produce acetate from H2 + CO2 exist whereas methanogens that use the same substrates in a reaction with a similar free-energy yield do not.
Figures


References
-
- Bakker E P. Cell K+ and K+ transport systems in prokaryotes. In: Bakker E P, editor. Alkali cation transport systems in prokaryotes. Boca Raton, Fla: CRC Press, Inc.; 1993. pp. 205–224.
-
- Bakker E P. Low affinity K+ uptake systems. In: Bakker E P, editor. Alkali cation transport systems in prokaryotes. Boca Raton, Fla: CRC Press, Inc.; 1993. pp. 253–276.
-
- Bauchop T, Elsden S R. The growth of micro-organisms in relation to their energy supply. J Gen Microbiol. 1960;23:457–469. - PubMed
-
- Baxter R M, Gibbons N E. Effects of sodium and potassium chloride on certain enzymes of Micrococcus halodenitrificans and Pseudomonas salinaria. Can J Microbiol. 1956;2:599–606. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

