Microtubule-based endoplasmic reticulum motility in Xenopus laevis: activation of membrane-associated kinesin during development
- PMID: 10359605
- PMCID: PMC25389
- DOI: 10.1091/mbc.10.6.1909
Microtubule-based endoplasmic reticulum motility in Xenopus laevis: activation of membrane-associated kinesin during development
Abstract
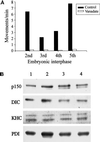
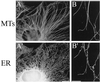
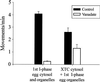
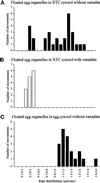
The endoplasmic reticulum (ER) in animal cells uses microtubule motor proteins to adopt and maintain its extended, reticular organization. Although the orientation of microtubules in many somatic cell types predicts that the ER should move toward microtubule plus ends, motor-dependent ER motility reconstituted in extracts of Xenopus laevis eggs is exclusively a minus end-directed, cytoplasmic dynein-driven process. We have used Xenopus egg, embryo, and somatic Xenopus tissue culture cell (XTC) extracts to study ER motility during embryonic development in Xenopus by video-enhanced differential interference contrast microscopy. Our results demonstrate that cytoplasmic dynein is the sole motor for microtubule-based ER motility throughout the early stages of development (up to at least the fifth embryonic interphase). When egg-derived ER membranes were incubated in somatic XTC cytosol, however, ER tubules moved in both directions along microtubules. Data from directionality assays suggest that plus end-directed ER tubule extensions contribute approximately 19% of the total microtubule-based ER motility under these conditions. In XTC extracts, the rate of ER tubule extensions toward microtubule plus ends is lower ( approximately 0.4 microm/s) than minus end-directed motility ( approximately 1.3 microm/s), and plus end-directed motility is eliminated by a function-blocking anti-conventional kinesin heavy chain antibody (SUK4). In addition, we provide evidence that the initiation of plus end-directed ER motility in somatic cytosol is likely to occur via activation of membrane-associated kinesin.
Figures




Similar articles
-
Role of kinesin-1 and cytoplasmic dynein in endoplasmic reticulum movement in VERO cells.J Cell Sci. 2009 Jun 15;122(Pt 12):1979-89. doi: 10.1242/jcs.041962. Epub 2009 May 19. J Cell Sci. 2009. PMID: 19454478 Free PMC article.
-
Cell cycle regulation of dynein association with membranes modulates microtubule-based organelle transport.J Cell Biol. 1996 May;133(3):585-93. doi: 10.1083/jcb.133.3.585. J Cell Biol. 1996. PMID: 8636233 Free PMC article.
-
Kinesin is the motor for microtubule-mediated Golgi-to-ER membrane traffic.J Cell Biol. 1995 Feb;128(3):293-306. doi: 10.1083/jcb.128.3.293. J Cell Biol. 1995. PMID: 7844144 Free PMC article.
-
Dynamic phase microscopy reveals periodic oscillations of endoplasmic reticulum during network formation.Biochemistry (Mosc). 2014 Sep;79(9):907-16. doi: 10.1134/S0006297914090077. Biochemistry (Mosc). 2014. PMID: 25385018 Review.
-
Isolation and analysis of microtubule motor proteins.Methods Cell Biol. 1994;44:279-88. doi: 10.1016/s0091-679x(08)60919-x. Methods Cell Biol. 1994. PMID: 7707957 Review.
Cited by
-
Mitotic inheritance of endoplasmic reticulum in the primitive red alga Cyanidioschyzon merolae.Protoplasma. 2012 Oct;249(4):1129-35. doi: 10.1007/s00709-011-0359-1. Epub 2011 Dec 13. Protoplasma. 2012. PMID: 22160190
-
Dynein supports motility of endoplasmic reticulum in the fungus Ustilago maydis.Mol Biol Cell. 2002 Mar;13(3):965-77. doi: 10.1091/mbc.01-10-0475. Mol Biol Cell. 2002. PMID: 11907275 Free PMC article.
-
Multiple kinesin motors coordinate cytoplasmic RNA transport on a subpopulation of microtubules in Xenopus oocytes.Dev Cell. 2008 Sep;15(3):426-436. doi: 10.1016/j.devcel.2008.06.014. Epub 2008 Sep 4. Dev Cell. 2008. PMID: 18771961 Free PMC article.
-
The interconnection of endoplasmic reticulum and microtubule and its implication in Hereditary Spastic Paraplegia.Comput Struct Biotechnol J. 2023 Feb 20;21:1670-1677. doi: 10.1016/j.csbj.2023.02.025. eCollection 2023. Comput Struct Biotechnol J. 2023. PMID: 36860342 Free PMC article. Review.
-
Apoptotic cleavage of cytoplasmic dynein intermediate chain and p150(Glued) stops dynein-dependent membrane motility.J Cell Biol. 2001 Jun 25;153(7):1415-26. doi: 10.1083/jcb.153.7.1415. J Cell Biol. 2001. PMID: 11425872 Free PMC article.
References
-
- Allan V. Assay of membrane motility in interphase and metaphase Xenopus extracts. In: Scholey JM, editor. Methods in Cell Biology. San Diego: Academic Press; 1993. pp. 203–226. - PubMed
-
- Allan V. Role of motor proteins in organizing the endoplasmic reticulum and Golgi apparatus. Semin Cell Dev Biol. 1996;7:335–342.
-
- Allan V, Vale R. Movement of membrane tubules along microtubules in vitro: evidence for specialized sites of motor attachment. J Cell Sci. 1994;107:1885–1897. - PubMed
-
- Allan VJ. Organelle motility and membrane network formation in metaphase and interphase cell-free extracts. Methods Enzymol. 1998;298:339–353. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

