Calcitriol directly sensitizes renal tubular cells to ATP-depletion- and iron-mediated attack
- PMID: 10362817
- PMCID: PMC1866639
- DOI: 10.1016/S0002-9440(10)65448-X
Calcitriol directly sensitizes renal tubular cells to ATP-depletion- and iron-mediated attack
Abstract
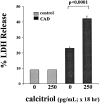
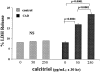
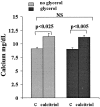
Vitamin Ds have been reported to have diverse effects on cell homeostasis, leading to suggestions that they have therapeutic applications extending beyond their traditional actions on the Ca2+/parathyroid/bone axis. As some of these potential indications carry an inherent risk of acute renal failure (ARF; eg, cancer chemotherapy and organ transplantation), the goal of this study was to assess whether vitamin Ds directly affect renal tubule injury responses. Cultured human proximal tubular (HK-2) cells were exposed to physiological or pharmacological doses of either calcitriol (D3) or a synthetic vitamin D2 analogue (19-nor) for 3 to 48 hours. Their impact on cell integrity (percent lactate dehydrogenase (LDH) release and tetrazolium dye MTT uptake) under basal conditions and during superimposed injuries (ATP depletion/Ca2+ ionophore or iron-mediated oxidant stress) were determined. As vitamin Ds can be anti-proliferative, cell outgrowth ([3H]thymidine uptake and crystal violet staining) was also tested. Finally, the action of D3 on in vivo ARF (glycerol-induced myoglobinuria) and isolated proximal tubule injury responses were assessed. D3 induced a rapid, dose-dependent increase in HK-2 susceptibility to both ATP-depletion/Ca2+-ionophore- and Fe-mediated attack without independently affecting cell integrity or proliferative responses. In contrast, D2 negatively affected only Fe toxicity and only after relatively prolonged exposure (48 hours). D3 dramatically potentiated in vivo ARF (two- to threefold increase in azotemia), suggesting potential in vivo relevance of the above HK-2 cell results. Proximal tubules, isolated from these glycerol-exposed mice, suggested that D3 can worsen tubule injury despite a parodoxic suppression of H2O2 production. In contrast, D3 had a mild negative impact on cellular energetics (depressed ATP/ADP ratios), and it accentuated plasma membrane phospholipid breakdown. The latter was observed in both glycerol-treated and control tubules, suggesting a primary role in the injury- potentiation effect of D3. Vitamins D(s) may directly, and differentially, increase proximal tubule cell susceptibility to superimposed attack. This property should be considered as new uses for these agents are defined.
Figures

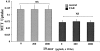
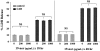
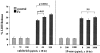
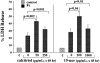
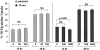
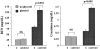
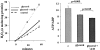
Similar articles
-
Plasma membrane cholesterol: a critical determinant of cellular energetics and tubular resistance to attack.Kidney Int. 2000 Jul;58(1):193-205. doi: 10.1046/j.1523-1755.2000.00154.x. Kidney Int. 2000. PMID: 10886564
-
Polyene antibiotics: relative degrees of in vitro cytotoxicity and potential effects on tubule phospholipid and ceramide content.Am J Kidney Dis. 2000 Aug;36(2):238-49. doi: 10.1053/ajkd.2000.8967. Am J Kidney Dis. 2000. PMID: 10922301
-
Sphingomyelinase and membrane sphingomyelin content: determinants ofProximal tubule cell susceptibility to injury.J Am Soc Nephrol. 2000 May;11(5):894-902. doi: 10.1681/ASN.V115894. J Am Soc Nephrol. 2000. PMID: 10770967
-
Mechanisms of cell injury in ATP-depleted proximal tubules. Role of glycine, calcium, and polyphosphoinositides.Nephrol Dial Transplant. 1994;9 Suppl 4:15-21. Nephrol Dial Transplant. 1994. PMID: 7800249 Review.
-
1alpha(OH)D3 One-alpha-hydroxy-cholecalciferol--an active vitamin D analog. Clinical studies on prophylaxis and treatment of secondary hyperparathyroidism in uremic patients on chronic dialysis.Dan Med Bull. 2008 Nov;55(4):186-210. Dan Med Bull. 2008. PMID: 19232159 Review.
Cited by
-
Paricalcitol: a review of its use in the management of secondary hyperparathyroidism.Drugs. 2005;65(4):559-76. doi: 10.2165/00003495-200565040-00008. Drugs. 2005. PMID: 15733015 Review.
-
Changes in free and esterified cholesterol: hallmarks of acute renal tubular injury and acquired cytoresistance.Am J Pathol. 2000 Sep;157(3):1007-16. doi: 10.1016/S0002-9440(10)64613-5. Am J Pathol. 2000. PMID: 10980139 Free PMC article.
References
-
- DeLuca HF, Zierold C: Mechanisms and functions of vitamin D. Nutr Rev 1998, 56:S4-S10 - PubMed
-
- Stumpf WE, Sar M, DeLuca HF: Sites of action of 1,25 (OH)2 vitamin D3 identified by thaw-mount autoradiography. Cohn DV Talmage RV Mathews JL eds. Hormonal Control of Calcium Metabolism. 1981, :pp 22-229 Exerpta Medica Amsterdam, The Netherlands
-
- Stumpf WE, Sar M, Reid FA, Tanaka Y, DeLuca HF: Target cells for 1,25 (OH)2-dihydroxyvitamin D3 in intestinal tract, stomach, kidney, skin, pituitary, and parathyroid. Science 1979, 206:1188-1190 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

