Porphyrin-mediated binding to hemoglobin by the HA2 domain of cysteine proteinases (gingipains) and hemagglutinins from the periodontal pathogen Porphyromonas gingivalis
- PMID: 10368154
- PMCID: PMC93857
- DOI: 10.1128/JB.181.12.3784-3791.1999
Porphyrin-mediated binding to hemoglobin by the HA2 domain of cysteine proteinases (gingipains) and hemagglutinins from the periodontal pathogen Porphyromonas gingivalis
Abstract
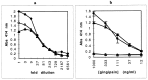
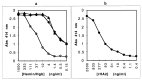
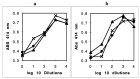
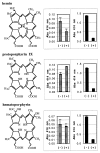
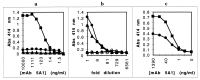
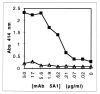
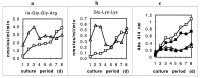
Heme binding and uptake are considered fundamental to the growth and virulence of the gram-negative periodontal pathogen Porphyromonas gingivalis. We therefore examined the potential role of the dominant P. gingivalis cysteine proteinases (gingipains) in the acquisition of heme from the environment. A recombinant hemoglobin-binding domain that is conserved between two predominant gingipains (domain HA2) demonstrated tight binding to hemin (Kd = 16 nM), and binding was inhibited by iron-free protoporphyrin IX (Ki = 2.5 microM). Hemoglobin binding to the gingipains and the recombinant HA2 (rHA2) domain (Kd = 2.1 nM) was also inhibited by protoporphyrin IX (Ki = 10 microM), demonstrating an essential interaction between the HA2 domain and the heme moiety in hemoglobin binding. Binding of rHA2 with either hemin, protoporphyrin IX, or hematoporphyrin was abolished by establishing covalent linkage of the protoporphyrin propionic acid side chains to fixed amines, demonstrating specific and directed binding of rHA2 to these protoporphyrins. A monoclonal antibody which recognizes a peptide epitope within the HA2 domain was employed to demonstrate that HA2-associated hemoglobin-binding activity was expressed and released by P. gingivalis cells in a batch culture, in parallel with proteinase activity. Cysteine proteinases from P. gingivalis appear to be multidomain proteins with functions for hemagglutination, erythrocyte lysis, proteolysis, and heme binding, as demonstrated here. Detailed understanding of the biochemical pathways for heme acquisition in P. gingivalis may allow precise targeting of this critical metabolic aspect for periodontal disease prevention.
Figures

Similar articles
-
Binding specificity of the Porphyromonas gingivalis heme and hemoglobin receptor HmuR, gingipain K, and gingipain R1 for heme, porphyrins, and metalloporphyrins.J Bacteriol. 2001 Oct;183(19):5599-608. doi: 10.1128/JB.183.19.5599-5608.2001. J Bacteriol. 2001. PMID: 11544222 Free PMC article.
-
Porphyrin-mediated cell surface heme capture from hemoglobin by Porphyromonas gingivalis.J Bacteriol. 2003 Apr;185(8):2528-37. doi: 10.1128/JB.185.8.2528-2537.2003. J Bacteriol. 2003. PMID: 12670977 Free PMC article.
-
Identification of amino acid residues involved in hemin binding in Porphyromonas gingivalis hemagglutinin 2.Mol Oral Microbiol. 2015 Oct;30(5):337-46. doi: 10.1111/omi.12097. Epub 2015 May 11. Mol Oral Microbiol. 2015. PMID: 25833325
-
Porphyromonas gingivalis gingipains: the molecular teeth of a microbial vampire.Curr Protein Pept Sci. 2003 Dec;4(6):409-26. doi: 10.2174/1389203033487009. Curr Protein Pept Sci. 2003. PMID: 14683427 Review.
-
Role of gingipains R in the pathogenesis of Porphyromonas gingivalis-mediated periodontal disease.Clin Infect Dis. 1999 Mar;28(3):456-65. doi: 10.1086/515156. Clin Infect Dis. 1999. PMID: 10194062 Review.
Cited by
-
Functional implication of the hydrolysis of platelet endothelial cell adhesion molecule 1 (CD31) by gingipains of Porphyromonas gingivalis for the pathology of periodontal disease.Infect Immun. 2005 Mar;73(3):1386-98. doi: 10.1128/IAI.73.3.1386-1398.2005. Infect Immun. 2005. PMID: 15731036 Free PMC article.
-
The hemoglobin receptor protein of porphyromonas gingivalis inhibits receptor activator NF-kappaB ligand-induced osteoclastogenesis from bone marrow macrophages.Infect Immun. 2006 May;74(5):2544-51. doi: 10.1128/IAI.74.5.2544-2551.2006. Infect Immun. 2006. PMID: 16622189 Free PMC article.
-
Identification of amino acid residues involved in heme binding and hemoprotein utilization in the Porphyromonas gingivalis heme receptor HmuR.Infect Immun. 2006 Feb;74(2):1222-32. doi: 10.1128/IAI.74.2.1222-1232.2006. Infect Immun. 2006. PMID: 16428772 Free PMC article.
-
An early report: a modified porphyrin-linked metronidazole targeting intracellular Porphyromonas gingivalis in cultured oral epithelial cells.Int J Oral Sci. 2017 Sep;9(3):167-173. doi: 10.1038/ijos.2017.31. Int J Oral Sci. 2017. PMID: 28960193 Free PMC article.
-
Binding specificity of the Porphyromonas gingivalis heme and hemoglobin receptor HmuR, gingipain K, and gingipain R1 for heme, porphyrins, and metalloporphyrins.J Bacteriol. 2001 Oct;183(19):5599-608. doi: 10.1128/JB.183.19.5599-5608.2001. J Bacteriol. 2001. PMID: 11544222 Free PMC article.
References
-
- Albandar J M, Brown L J, Löe H. Putative periodontal pathogens in subgingival plaque of young adults with and without early-onset periodontitis. J Periodontol. 1997;68:973–981. - PubMed
-
- Beck J D, Offenbacher S, Williams R, Gibbs P, Garcia R. Periodontitis: a risk factor for coronary heart disease? Ann Periodontol. 1998;3:127–141. - PubMed
-
- Bedi G S, Williams T. Purification and characterization of a collagen-degrading protease from Porphyromonas gingivalis. J Biol Chem. 1994;269:599–606. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

