Genotype, phenotype, and protein structure in a regulator of sporulation: effects of mutations in the spoIIAA gene of Bacillus subtilis
- PMID: 10368168
- PMCID: PMC93871
- DOI: 10.1128/JB.181.12.3860-3863.1999
Genotype, phenotype, and protein structure in a regulator of sporulation: effects of mutations in the spoIIAA gene of Bacillus subtilis
Abstract
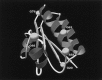
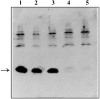
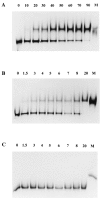
SpoIIAA, a phosphorylatable protein, is essential to the regulation of sigmaF, the first sporulation-specific transcription factor of Bacillus subtilis. The solution structure of SpoIIAA has recently been published. Here we examine four mutant SpoIIAA proteins and correlate their properties with the phenotypes of the corresponding B. subtilis mutant strains. Two of the mutations severely disrupted the structure of the protein, a third greatly diminished the rate of its phosphorylation and abolished dephosphorylation, and the fourth left phosphorylation unaffected but reduced the rate of dephosphorylation about 10-fold.
Figures

Similar articles
-
Bacillus subtilis mutations that alter the pathway of phosphorylation of the anti-anti-sigmaF factor SpoIIAA lead to a Spo- phenotype.Mol Microbiol. 2001 Apr;40(1):9-19. doi: 10.1046/j.1365-2958.2001.02353.x. Mol Microbiol. 2001. PMID: 11298272
-
The kinase activity of the antisigma factor SpoIIAB is required for activation as well as inhibition of transcription factor sigmaF during sporulation in Bacillus subtilis.J Mol Biol. 1998 Dec 4;284(3):569-78. doi: 10.1006/jmbi.1998.2202. J Mol Biol. 1998. PMID: 9826499
-
Efficient regulation of sigmaF, the first sporulation-specific sigma factor in B.subtilis.J Mol Biol. 2004 Sep 24;342(4):1187-95. doi: 10.1016/j.jmb.2004.07.090. J Mol Biol. 2004. PMID: 15351644
-
Control of the cell-specificity of sigma F activity in Bacillus subtilis.Philos Trans R Soc Lond B Biol Sci. 1996 Apr 29;351(1339):537-42. doi: 10.1098/rstb.1996.0052. Philos Trans R Soc Lond B Biol Sci. 1996. PMID: 8735276 Review.
-
Differential gene expression in genetically identical sister cells: the initiation of sporulation in Bacillus subtilis.Mol Microbiol. 2005 May;56(3):578-89. doi: 10.1111/j.1365-2958.2005.04594.x. Mol Microbiol. 2005. PMID: 15819616 Review.
Cited by
-
NMR studies of the sporulation protein SpoIIAA: implications for the regulation of the transcription factor sigmaF in Bacillus subtilis.J Biomol NMR. 2001 Apr;19(4):293-304. doi: 10.1023/a:1011247523112. J Biomol NMR. 2001. PMID: 11370776
References
-
- Alper S, Duncan L, Losick R. An adenosine nucleotide switch controlling the activity of a cell type-specific transcription factor in B. subtilis. Cell. 1994;77:195–205. - PubMed
-
- Decatur A L, Losick R. Three sites of contact between the Bacillus subtilis transcription factor ςFand its antisigma factor SpoIIAB. Genes Dev. 1996;10:2348–2358. - PubMed
-
- Diederich B, Wilkinson J F, Magnin T, Najafi S M A, Errington J, Yudkin M D. Role of interactions between SpoIIAA and SpoIIAB in regulating cell-specific transcription factor ςF of Bacillus subtilis. Genes Dev. 1994;8:2653–2663. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

