Upstream stimulatory factor regulates major histocompatibility complex class I gene expression: the U2DeltaE4 splice variant abrogates E-box activity
- PMID: 10373528
- PMCID: PMC84277
- DOI: 10.1128/MCB.19.7.4788
Upstream stimulatory factor regulates major histocompatibility complex class I gene expression: the U2DeltaE4 splice variant abrogates E-box activity
Abstract
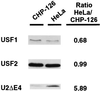
The tissue-specific expression of major histocompatibility complex class I genes is determined by a series of upstream regulatory elements, many of which remain ill defined. We now report that a distal E-box element, located between bp -309 and -314 upstream of transcription initiation, acts as a cell type-specific enhancer of class I promoter activity. The class I E box is very active in a neuroblastoma cell line, CHP-126, but is relatively inactive in the HeLa epithelial cell line. The basic helix-loop-helix leucine zipper proteins upstream stimulatory factor 1 (USF1) and USF2 were shown to specifically recognize the class I E box, resulting in the activation of the downstream promoter. Fine mapping of USF1 and USF2 amino-terminal functional domains revealed differences in their abilities to activate the class I E box. Whereas USF1 contained only an extended activation domain, USF2 contained both an activation domain and a negative regulatory region. Surprisingly, the naturally occurring splice variant of USF2 lacking the exon 4 domain, U2DeltaE4, acted as a dominant-negative regulator of USF-mediated activation of the class I promoter. This latter activity is in sharp contrast to the known ability of U2DeltaE4 to activate the adenovirus major late promoter. Class I E-box function is correlated with the relative amount of U2DeltaE4 in a cell, leading to the proposal that U2DeltaE4 modulates class I E-box activity and may represent one mechanism to fine-tune class I expression in various tissues.
Figures




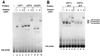



Similar articles
-
Upstream stimulatory factors, USF1 and USF2, bind to the human haem oxygenase-1 proximal promoter in vivo and regulate its transcription.Biochem J. 2004 Oct 15;383(Pt 2):209-18. doi: 10.1042/BJ20040794. Biochem J. 2004. PMID: 15242350 Free PMC article.
-
Regulation of the nuclear gene that encodes the alpha-subunit of the mitochondrial F0F1-ATP synthase complex. Activation by upstream stimulatory factor 2.J Biol Chem. 1997 Apr 18;272(16):10538-42. doi: 10.1074/jbc.272.16.10538. J Biol Chem. 1997. PMID: 9099698
-
The basic helix-loop-helix, leucine zipper transcription factor, USF (upstream stimulatory factor), is a key regulator of SF-1 (steroidogenic factor-1) gene expression in pituitary gonadotrope and steroidogenic cells.Mol Endocrinol. 1998 May;12(5):714-26. doi: 10.1210/mend.12.5.0100. Mol Endocrinol. 1998. PMID: 9605934
-
Upstream stimulatory factors 1 and 2 mediate the transcription of angiotensin II binding and inhibitory protein.J Biol Chem. 2013 Jun 28;288(26):19238-49. doi: 10.1074/jbc.M113.451054. Epub 2013 May 7. J Biol Chem. 2013. PMID: 23653383 Free PMC article.
-
Human prolyl-4-hydroxylase alpha(I) transcription is mediated by upstream stimulatory factors.J Biol Chem. 2006 Apr 21;281(16):10849-55. doi: 10.1074/jbc.M511237200. Epub 2006 Feb 17. J Biol Chem. 2006. PMID: 16488890 Free PMC article.
Cited by
-
Distinct transcriptional pathways regulate basal and activated major histocompatibility complex class I expression.Mol Cell Biol. 2003 May;23(10):3377-91. doi: 10.1128/MCB.23.10.3377-3391.2003. Mol Cell Biol. 2003. PMID: 12724398 Free PMC article.
-
The USF proteins regulate transcription of the follicle-stimulating hormone receptor but are insufficient for cell-specific expression.Mol Endocrinol. 2000 Nov;14(11):1836-48. doi: 10.1210/mend.14.11.0557. Mol Endocrinol. 2000. PMID: 11075816 Free PMC article.
-
The role of upstream stimulatory factor 1 in the transcriptional regulation of the human TBX21 promoter mediated by the T-1514C polymorphism associated with systemic lupus erythematosus.Immunogenetics. 2012 May;64(5):361-70. doi: 10.1007/s00251-011-0597-6. Epub 2012 Jan 19. Immunogenetics. 2012. PMID: 22258560
-
Canonical and single-cell Hi-C reveal distinct chromatin interaction sub-networks of mammalian transcription factors.Genome Biol. 2018 Oct 25;19(1):174. doi: 10.1186/s13059-018-1558-2. Genome Biol. 2018. PMID: 30359306 Free PMC article.
-
Three novel downstream promoter elements regulate MHC class I promoter activity in mammalian cells.PLoS One. 2010 Dec 13;5(12):e15278. doi: 10.1371/journal.pone.0015278. PLoS One. 2010. PMID: 21179443 Free PMC article.
References
-
- Aperlo C, Boulukos K E, Pognonec P. The basic region/helix-loop-helix/leucine repeat transcription factor USF interferes with Ras transformation. Eur J Biochem. 1996;241:249–253. - PubMed
-
- Beckman H, Su L-K, Kadesch T. TFE3: a helix-loop-helix protein that activates transcription through the immunoglobulin enhancer uE3 motif. Genes Dev. 1989;4:1730–1740. - PubMed
-
- Bodor J, Habener J F. Role of transcriptional repressor ICER in cyclic AMP-mediated attenuation of cytokine gene expression in human thymocytes. J Biol Chem. 1998;273:9544–9551. - PubMed
-
- Bresnick E H, Felsenfeld G. The leucine zipper is necessary for stabilizing a dimer of the helix-loop-helix transcription factor USF but not for maintenance of an elongated conformation. J Biol Chem. 1994;269:21110–21116. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
