SOCS-3 is tyrosine phosphorylated in response to interleukin-2 and suppresses STAT5 phosphorylation and lymphocyte proliferation
- PMID: 10373548
- PMCID: PMC84319
- DOI: 10.1128/MCB.19.7.4980
SOCS-3 is tyrosine phosphorylated in response to interleukin-2 and suppresses STAT5 phosphorylation and lymphocyte proliferation
Abstract
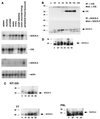
Members of the recently discovered SOCS/CIS/SSI family have been proposed as regulators of cytokine signaling, and while targets and mechanisms have been suggested for some family members, the precise role of these proteins remains to be defined. To date no SOCS proteins have been specifically implicated in interleukin-2 (IL-2) signaling in T cells. Here we report SOCS-3 expression in response to IL-2 in both T-cell lines and human peripheral blood lymphocytes. SOCS-3 protein was detectable as early as 30 min following IL-2 stimulation, while CIS was seen only at low levels after 2 h. Unlike CIS, SOCS-3 was rapidly tyrosine phosphorylated in response to IL-2. Tyrosine phosphorylation of SOCS-3 was observed upon coexpression with Jak1 and Jak2 but only weakly with Jak3. In these experiments, SOCS-3 associated with Jak1 and inhibited Jak1 phosphorylation, and this inhibition was markedly enhanced by the presence of IL-2 receptor beta chain (IL-2Rbeta). Moreover, following IL-2 stimulation of T cells, SOCS-3 was able to interact with the IL-2 receptor complex, and in particular tyrosine phosphorylated Jak1 and IL-2Rbeta. Additionally, in lymphocytes expressing SOCS-3 but not CIS, IL-2-induced tyrosine phosphorylation of STAT5b was markedly reduced, while there was only a weak effect on IL-3-mediated STAT5b tyrosine phosphorylation. Finally, proliferation induced by both IL-2- and IL-3 was significantly inhibited in the presence of SOCS-3. The findings suggest that when SOCS-3 is rapidly induced by IL-2 in T cells, it acts to inhibit IL-2 responses in a classical negative feedback loop.
Figures



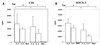
References
-
- Adams T E, Hansen J A, Starr R, Nicola N A, Hilton D J, Billestrup N. Growth hormone preferentially induces the rapid, transient expression of SOCS-3, a novel inhibitor of cytokine receptor signaling. J Biol Chem. 1998;273:1285–1287. - PubMed
-
- Auernhammer C J, Chesnokova V, Bousquet C, Melmed S. Pituitary corticotroph SOCS-3: novel intracellular regulation of leukemia-inhibitory factor-mediated proopiomelanocortin gene expression and adrenocorticotropin secretion. Mol Endocrinol. 1998;12:954–961. - PubMed
-
- Bjorbaek C, Elmquist J K, Frantz J D, Shoelson S E, Flier J S. Identification of SOCS-3 as a potential mediator of central leptin resistance. Mol Cell. 1998;1:619–625. - PubMed
-
- Callus B A, Mathey-Prevot B. Interleukin-3-induced activation of the JAK/STAT pathway is prolonged by proteasome inhibitors. Blood. 1998;91:3182–3192. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
