Long-term behavioral effects of repetitive pain in neonatal rat pups
- PMID: 10386907
- PMCID: PMC4211637
- DOI: 10.1016/s0031-9384(98)00338-2
Long-term behavioral effects of repetitive pain in neonatal rat pups
Abstract
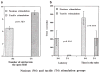
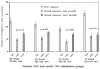
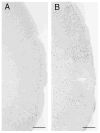
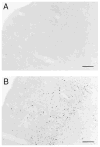
Human preterm neonates are subjected to repetitive pain during neonatal intensive care. We hypothesized that exposure to repetitive neonatal pain may cause permanent or long-term changes because of the developmental plasticity of the immature brain. Neonatal rat pups were stimulated one, two, or four times each day from P0 to P7 with either needle prick (noxious groups N1, N2, N4) or cotton tip rub (tactile groups T1, T2, T4). In groups N2, N4, T2, T4 stimuli were applied to separate paws at hourly intervals;each paw was stimulated only once a day. Identical rearing occurred from P7 to P22 days. Pain thresholds were measured on P16, P22, and P65 (hot-plate test), and testing for defensive withdrawal, alcohol preference, air-puff startle, and social discrimination tests occurred during adulthood. Adult rats were exposed to a hot plate at 62 degrees C for 20 s, then sacrificed and perfused at 0 and 30 min after exposure. Fos expression in the somatosensory cortex was measured by immunocytochemistry. Weight gain in the N2 group was greater than the T2 group on P16 (p < 0.05) and P22 (p < 0.005); no differences occurred in the other groups. Decreased pain latencies were noted in the N4 group [5.0 +/- 1.0 s vs. 6.2 +/- 1.4 s on P16 (p < 0.05); 3.9 +/- 0.5 s vs. 5.5 +/- 1.6 s on P22 (p < 0.005)], indicating effects of repetitive neonatal pain on subsequent development of the pain system. As adults, N4 group rats showed an increased preference for alcohol (55 +/- 18% vs. 32 +/- 21%; p = 0.004); increased latency in exploratory and defensive withdrawal behavior (p < 0.05); and a prolonged chemosensory memory in the social discrimination test (p < 0.05). No significant differences occurred in corticosterone and ACTH levels following air-puff startle or in pain thresholds at P65 between N4 and T4 groups. Fos expression at 30 min after hot-plate exposure was significantly greater in all areas of the somatosensory cortex in the T4 group compared with the N4 group (p < 0.05), whereas no differences occurred just after exposure. These data suggest that repetitive pain in neonatal rat pups may lead to an altered development of the pain system associated with decreased pain thresholds during development. Increased plasticity of the neonatal brain may allow these and other changes in brain development to increase their vulnerability to stress disorders and anxiety-mediated adult behavior. Similar behavioral changes have been observed during the later childhood of expreterm neonates who were exposed to prolonged periods of neonatal intensive care.
Figures



References
-
- Adams N, Oldham TD. Seminatural housing increases subsequent ethanol intake in male Maudsley Reactive rats. J Stud Alcohol. 1996;57:349–351. - PubMed
-
- Adams N. Sex differences and the effects of tail pinch on ethanol drinking in Maudsley rats. Alcohol. 1995;12:463–468. - PubMed
-
- Badishtov BA, Overstreet DH, Kashevskaya OP, et al. To drink or not to drink: Open field behavior in alcohol-preferring and nonpreferring rat strains. Physiol Behav. 1995;57:585–589. - PubMed
-
- Bergvall AH, Fahike CJ, Aonsson L, Hansen S. In quest for a possible association between heightened social aggression and excessive alcohol drinking in the rat. Physiol Behav. 1996;59:807–812. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

