Design, total chemical synthesis, and binding properties of a [Leu-91-N1-methyl-7-azaTrp]Ras-binding domain of c-Raf-1
- PMID: 10393913
- PMCID: PMC22153
- DOI: 10.1073/pnas.96.14.7865
Design, total chemical synthesis, and binding properties of a [Leu-91-N1-methyl-7-azaTrp]Ras-binding domain of c-Raf-1
Abstract
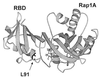
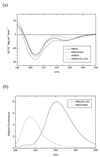
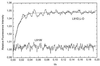
The Ras-binding domain (RBD) of c-Raf-1 has been synthesized chemically, taking advantage of the chemical ligation of two peptide fragments of the protein. This procedure allowed incorporation of an unnatural amino acid (N1-methyl-7-azatryptophan) at position 91 of RBD, producing a protein with fluorescent properties distinct from and distinguishable from those of proteins containing the natural fluorophore tryptophan. The resulting protein was shown to interact with Ras in a manner that was almost indistinguishable from that of unmodified RBD based on transient kinetic monitoring of the binding event. Modified RBD containing the L-isomer of the unnatural amino acid or its racemic D,L mixture appeared to interact identically with Ras. The approach demonstrates a general procedure for the introduction of unnatural amino acids that can be used for monitoring protein-protein interactions and for the introduction of an unnatural backbone structure at strategic positions.
Figures


Similar articles
-
Total chemical synthesis of a functional interacting protein pair: the protooncogene H-Ras and the Ras-binding domain of its effector c-Raf1.Proc Natl Acad Sci U S A. 2003 Apr 29;100(9):5075-80. doi: 10.1073/pnas.0831227100. Epub 2003 Apr 18. Proc Natl Acad Sci U S A. 2003. PMID: 12704243 Free PMC article.
-
A sensitive fluorescence monitor for the detection of activated Ras: total chemical synthesis of site-specifically labeled Ras binding domain of c-Raf1 immobilized on a surface.Chem Biol. 2001 Mar;8(3):243-52. doi: 10.1016/s1074-5521(01)00003-5. Chem Biol. 2001. PMID: 11306349
-
The transition state of the ras binding domain of Raf is structurally polarized based on Phi-values but is energetically diffuse.J Mol Biol. 2007 Feb 2;365(5):1559-77. doi: 10.1016/j.jmb.2006.10.079. Epub 2006 Oct 28. J Mol Biol. 2007. PMID: 17137592
-
Meaningful relationships: the regulation of the Ras/Raf/MEK/ERK pathway by protein interactions.Biochem J. 2000 Oct 15;351 Pt 2(Pt 2):289-305. Biochem J. 2000. PMID: 11023813 Free PMC article. Review.
-
The RAF family: an expanding network of post-translational controls and protein-protein interactions.Cell Res. 1998 Jun;8(2):81-98. doi: 10.1038/cr.1998.9. Cell Res. 1998. PMID: 9669024 Review.
Cited by
-
Total chemical synthesis of a functional interacting protein pair: the protooncogene H-Ras and the Ras-binding domain of its effector c-Raf1.Proc Natl Acad Sci U S A. 2003 Apr 29;100(9):5075-80. doi: 10.1073/pnas.0831227100. Epub 2003 Apr 18. Proc Natl Acad Sci U S A. 2003. PMID: 12704243 Free PMC article.
-
Advances in the chemical synthesis of human proteoforms.Sci China Life Sci. 2025 Apr 8. doi: 10.1007/s11427-024-2860-5. Online ahead of print. Sci China Life Sci. 2025. PMID: 40210795 Review.
-
Total chemical synthesis and electrophysiological characterization of mechanosensitive channels from Escherichia coli and Mycobacterium tuberculosis.Proc Natl Acad Sci U S A. 2004 Apr 6;101(14):4764-9. doi: 10.1073/pnas.0305693101. Epub 2004 Mar 23. Proc Natl Acad Sci U S A. 2004. PMID: 15041744 Free PMC article.
References
-
- Marshall M S. FASEB J. 1995;9:1311–1318. - PubMed
-
- Leevers S J, Paterson H F, Marshall C J. Nature (London) 1994;369:411–414. - PubMed
-
- Stokoe D, Macdonald S G, Cadwallader K, Symons M, Hancock J F. Science. 1994;264:1463–1467. - PubMed
-
- Barbacid M. Annu Rev Biochem. 1987;56:779–827. - PubMed
-
- Vojtek A B, Hollenberg S M, Cooper J A. Cell. 1993;74:205–214. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

