Impairment of spermatogenesis in mice lacking a functional aromatase (cyp 19) gene
- PMID: 10393934
- PMCID: PMC22174
- DOI: 10.1073/pnas.96.14.7986
Impairment of spermatogenesis in mice lacking a functional aromatase (cyp 19) gene
Abstract
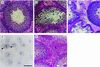
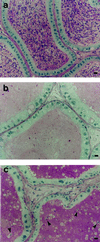
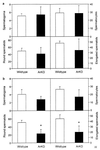
It is well established that spermatogenesis is controlled by gonadotrophins and testosterone. However, a role for estrogens in male reproduction recently was suggested in adult mice deficient in estrogen receptor alpha. These mice became infertile primarily because of an interruption of fluid reabsorption by the efferent ductules of the epididymis, thus leading to a disruption of the seminiferous epithelium [Hess, R. A., Bunick, D., Lee, K. H., Bahr, J., Taylor, J. A., Korach, K. S., and Lubahn, D. B. (1997) Nature (London) 390, 509-512]. Despite the demonstration of the aromatase enzyme, which converts androgens to estrogens, and estrogen receptors within the rodent seminiferous epithelium, the role of aromatase and estrogen in germ cell development is unknown. We have investigated spermatogenesis in mice that lack aromatase because of the targeted disruption of the cyp19 gene (ArKO). Male mice deficient in aromatase were initially fertile but developed progressive infertility, until their ability to sire pups was severely impaired. The mice deficient in aromatase developed disruptions to spermatogenesis between 4.5 months and 1 year, despite no decreases in gonadotrophins or androgens. Spermatogenesis primarily was arrested at early spermiogenic stages, as characterized by an increase in apoptosis and the appearance of multinucleated cells, and there was a significant reduction in round and elongated spermatids, but no changes in Sertoli cells and earlier germ cells. In addition, Leydig cell hyperplasia/hypertrophy was evident, presumably as a consequence of increased circulating luteinizing hormone. Our findings indicate that local expression of aromatase is essential for spermatogenesis and provide evidence for a direct action of estrogen on male germ cell development and thus fertility.
Figures

Similar articles
-
Targeted disruption of the estrogen receptor gene in male mice causes alteration of spermatogenesis and infertility.Endocrinology. 1996 Nov;137(11):4796-805. doi: 10.1210/endo.137.11.8895349. Endocrinology. 1996. PMID: 8895349
-
Genetic mutations resulting in estrogen insufficiency in the male.Mol Cell Endocrinol. 1998 Oct 25;145(1-2):55-9. doi: 10.1016/s0303-7207(98)00169-5. Mol Cell Endocrinol. 1998. PMID: 9922099 Review.
-
Aromatase expression and role of estrogens in male gonad : a review.Reprod Biol Endocrinol. 2003 Apr 11;1:35. doi: 10.1186/1477-7827-1-35. Reprod Biol Endocrinol. 2003. PMID: 12747806 Free PMC article. Review.
-
Aromatase expression in male germ cells.J Steroid Biochem Mol Biol. 2001 Dec;79(1-5):203-8. doi: 10.1016/s0960-0760(01)00137-6. J Steroid Biochem Mol Biol. 2001. PMID: 11850226 Review.
-
The phenotype of the aromatase knockout mouse reveals dietary phytoestrogens impact significantly on testis function.Endocrinology. 2002 Aug;143(8):2913-21. doi: 10.1210/endo.143.8.8957. Endocrinology. 2002. PMID: 12130556
Cited by
-
TEX11 modulates germ cell proliferation by competing with estrogen receptor β for the binding to HPIP.Mol Endocrinol. 2012 Apr;26(4):630-42. doi: 10.1210/me.2011-1263. Epub 2012 Mar 1. Mol Endocrinol. 2012. PMID: 22383461 Free PMC article.
-
Male germ cell apoptosis: regulation and biology.Philos Trans R Soc Lond B Biol Sci. 2010 May 27;365(1546):1501-15. doi: 10.1098/rstb.2009.0124. Philos Trans R Soc Lond B Biol Sci. 2010. PMID: 20403866 Free PMC article. Review.
-
Phenotyping male infertility in the mouse: how to get the most out of a 'non-performer'.Hum Reprod Update. 2010 Mar-Apr;16(2):205-24. doi: 10.1093/humupd/dmp032. Epub 2009 Sep 15. Hum Reprod Update. 2010. PMID: 19758979 Free PMC article. Review.
-
Estrogens in Human Male Gonadotropin Secretion and Testicular Physiology From Infancy to Late Puberty.Front Endocrinol (Lausanne). 2020 Feb 25;11:72. doi: 10.3389/fendo.2020.00072. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 32158430 Free PMC article. Review.
-
Morphological comparison of the testis and efferent ductules between wild-type and estrogen receptor alpha knockout mice during postnatal development.J Anat. 2009 Jun;214(6):916-25. doi: 10.1111/j.1469-7580.2009.01080.x. J Anat. 2009. PMID: 19538635 Free PMC article.
References
-
- Carani C, Qin K, Simoni M, Faustini-Fustini M, Serpente S, Boyd J, Korach K S, Simpson E R. N Engl J Med. 1997;337:91–95. - PubMed
-
- Bilezijian J P, Morishima A, Bell J, Grumbach M M. N Engl J Med. 1998;339:599–603. - PubMed
-
- Eddy E M, Washburn T F, Bunch D O, Goulding E H, Gladen B C, Lubahn D B, Korach K S. Endocrinology. 1996;137:4796–4815. - PubMed
-
- Fisher J S, Millar M R, Majdic G, Saunders P T, Fraser H M, Sharpe R M. J Endocrinol. 1997;153:485–495. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

