A potential mechanism underlying the increased susceptibility of individuals with a polymorphism in NAD(P)H:quinone oxidoreductase 1 (NQO1) to benzene toxicity
- PMID: 10393963
- PMCID: PMC22203
- DOI: 10.1073/pnas.96.14.8150
A potential mechanism underlying the increased susceptibility of individuals with a polymorphism in NAD(P)H:quinone oxidoreductase 1 (NQO1) to benzene toxicity
Abstract
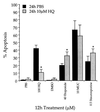
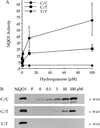
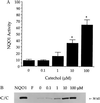
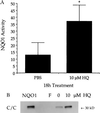
NAD(P)H:quinone oxidoreductase 1 (NQO1) is a two-electron reductase that detoxifies quinones derived from the oxidation of phenolic metabolites of benzene. A polymorphism in NQO1, a C609T substitution, has been identified, and individuals homozygous for this change (T/T) have no detectable NQO1. Exposed workers with a T/T genotype have an increased risk of benzene hematotoxicity. This finding suggests NQO1 is protective against benzene toxicity, which is difficult to reconcile with the lack of detectable NQO1 in human bone marrow. The human promyeloblastic cell line, KG-1a, was used to investigate the ability of the benzene metabolite hydroquinone (HQ) to induce NQO1. A concentration-dependent induction of NQO1 protein and activity was observed in KG-1a cells cultured with HQ. Multiple detoxification systems, including NQO1 and glutathione protect against benzene metabolite-induced toxicity. Indeed, exposure to a noncytotoxic concentration of HQ induced both NQO1 and soluble thiols and protected against HQ-induced apoptosis. NQO1 protein and activity increased in wild-type human bone marrow cells (C/C) exposed to HQ, whereas no NQO1 was induced by HQ in bone marrow cells with the T/T genotype. Intermediate induction of NQO1 by HQ was observed in heterozygous bone marrow cells (C/T). NQO1 also was induced by HQ in wild-type (C/C) human bone marrow CD34(+) progenitor cells. Our data suggest that failure to induce functional NQO1 may contribute to the increased risk of benzene poisoning in individuals homozygous for the NQO1 C609T substitution (T/T).
Figures



Comment in
-
Benzene, NQO1, and genetic susceptibility to cancer.Proc Natl Acad Sci U S A. 1999 Jul 6;96(14):7624-6. doi: 10.1073/pnas.96.14.7624. Proc Natl Acad Sci U S A. 1999. PMID: 10393869 Free PMC article. Review. No abstract available.
Similar articles
-
[Genetic polymorphisms of NQO1, GSTT1, GSTM1 and susceptibility to chronic benzene poisoning].Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi. 2005 Feb;23(1):1-5. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi. 2005. PMID: 15748501 Chinese.
-
Functions and distribution of NQO1 in human bone marrow: potential clues to benzene toxicity.Chem Biol Interact. 2005 May 30;153-154:137-46. doi: 10.1016/j.cbi.2005.03.018. Epub 2005 Apr 7. Chem Biol Interact. 2005. PMID: 15935810 Review.
-
Cell-specific activation and detoxification of benzene metabolites in mouse and human bone marrow: identification of target cells and a potential role for modulation of apoptosis in benzene toxicity.Environ Health Perspect. 1996 Dec;104 Suppl 6(Suppl 6):1177-82. doi: 10.1289/ehp.961041177. Environ Health Perspect. 1996. PMID: 9118890 Free PMC article.
-
Genotype-phenotype relationships in studies of a polymorphism in NAD(P)H:quinone oxidoreductase 1.Pharmacogenetics. 1999 Feb;9(1):113-21. doi: 10.1097/00008571-199902000-00015. Pharmacogenetics. 1999. PMID: 10208650
-
NAD(P)H:quinone oxidoreductase (NQO1) polymorphism, exposure to benzene, and predisposition to disease: a HuGE review.Genet Med. 2002 Mar-Apr;4(2):62-70. doi: 10.1097/00125817-200203000-00003. Genet Med. 2002. PMID: 11882782 Review.
Cited by
-
A functional NQO1 609C>T polymorphism and risk of gastrointestinal cancers: a meta-analysis.PLoS One. 2012;7(1):e30566. doi: 10.1371/journal.pone.0030566. Epub 2012 Jan 17. PLoS One. 2012. PMID: 22272361 Free PMC article.
-
Generation of stable ARE- driven reporter system for monitoring oxidative stress.Daru. 2015 Aug 1;23(1):38. doi: 10.1186/s40199-015-0122-9. Daru. 2015. PMID: 26231224 Free PMC article.
-
Benzene, NQO1, and genetic susceptibility to cancer.Proc Natl Acad Sci U S A. 1999 Jul 6;96(14):7624-6. doi: 10.1073/pnas.96.14.7624. Proc Natl Acad Sci U S A. 1999. PMID: 10393869 Free PMC article. Review. No abstract available.
-
Induction of murine NAD(P)H:quinone oxidoreductase by 2,3,7,8-tetrachlorodibenzo-p-dioxin requires the CNC (cap 'n' collar) basic leucine zipper transcription factor Nrf2 (nuclear factor erythroid 2-related factor 2): cross-interaction between AhR (aryl hydrocarbon receptor) and Nrf2 signal transduction.Biochem J. 2004 Jan 1;377(Pt 1):205-13. doi: 10.1042/BJ20031123. Biochem J. 2004. PMID: 14510636 Free PMC article.
-
Association of NQO1 polymorphism with spontaneous breast cancer in two independent populations.Br J Cancer. 2004 May 17;90(10):1989-94. doi: 10.1038/sj.bjc.6601779. Br J Cancer. 2004. PMID: 15138483 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

