Polymerizing microtubules activate site-directed F-actin assembly in nerve growth cones
- PMID: 10397767
- PMCID: PMC25445
- DOI: 10.1091/mbc.10.7.2309
Polymerizing microtubules activate site-directed F-actin assembly in nerve growth cones
Abstract
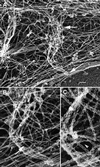
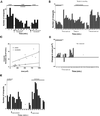
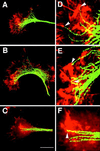
We identify an actin-based protrusive structure in growth cones termed "intrapodium." Unlike filopodia, intrapodia are initiated exclusively within lamellipodia and elongate in a continuous (nonsaltatory) manner parallel to the plane of the dorsal plasma membrane causing a ridge-like protrusion. Intrapodia resemble the actin-rich structures induced by intracellular pathogens (e.g., Listeria) or by extracellular beads. Cytochalasin B inhibits intrapodial elongation and removal of cytochalasin B produced a burst of intrapodial activity. Electron microscopic studies revealed that lamellipodial intrapodia contain both short and long actin filaments oriented with their barbed ends toward the membrane surface or advancing end. Our data suggest an interaction between microtubule endings and intrapodia formation. Disruption of microtubules by acute nocodazole treatment decreased intrapodia frequency, and washout of nocodazole or addition of the microtubule-stabilizing drug Taxol caused a burst of intrapodia formation. Furthermore, individual microtubule ends were found near intrapodia initiation sites. Thus, microtubule ends or associated structures may regulate these actin-dependent structures. We propose that intrapodia are the consequence of an early step in a cascade of events that leads to the development of F-actin-associated plasma membrane specializations.
Figures



Similar articles
-
Microtubule and Rac 1-dependent F-actin in growth cones.J Cell Sci. 2003 Sep 15;116(Pt 18):3739-48. doi: 10.1242/jcs.00686. Epub 2003 Jul 30. J Cell Sci. 2003. PMID: 12890754
-
Distribution of GAP-43, beta-III tubulin and F-actin in developing and regenerating axons and their growth cones in vitro, following neurotrophin treatment.J Neurocytol. 2003 Nov;32(9):1077-89. doi: 10.1023/B:NEUR.0000021903.24849.6c. J Neurocytol. 2003. PMID: 15044840
-
The cytoskeletons of isolated, neuronal growth cones.Neuroscience. 1987 Jun;21(3):977-89. doi: 10.1016/0306-4522(87)90052-2. Neuroscience. 1987. PMID: 2888041
-
Cytoskeletal social networking in the growth cone: How +TIPs mediate microtubule-actin cross-linking to drive axon outgrowth and guidance.Cytoskeleton (Hoboken). 2016 Sep;73(9):461-76. doi: 10.1002/cm.21272. Epub 2016 Feb 8. Cytoskeleton (Hoboken). 2016. PMID: 26783725 Free PMC article. Review.
-
Coupled zones of f-actin and microtubule movement in polarized cells.Dev Cell. 2002 Aug;3(2):152-3. doi: 10.1016/s1534-5807(02)00227-7. Dev Cell. 2002. PMID: 12194844 Review.
Cited by
-
Bridging the Gap: The Importance of TUBA1A α-Tubulin in Forming Midline Commissures.Front Cell Dev Biol. 2022 Jan 19;9:789438. doi: 10.3389/fcell.2021.789438. eCollection 2021. Front Cell Dev Biol. 2022. PMID: 35127710 Free PMC article.
-
Local Arp2/3-dependent actin assembly modulates applied traction force during apCAM adhesion site maturation.Mol Biol Cell. 2017 Jan 1;28(1):98-110. doi: 10.1091/mbc.E16-04-0228. Epub 2016 Nov 16. Mol Biol Cell. 2017. PMID: 27852899 Free PMC article.
-
Increase in Growth Cone Size Correlates with Decrease in Neurite Growth Rate.Neural Plast. 2016;2016:3497901. doi: 10.1155/2016/3497901. Epub 2016 May 4. Neural Plast. 2016. PMID: 27274874 Free PMC article.
-
Role of actin filaments in the axonal transport of microtubules.J Neurosci. 2004 Dec 15;24(50):11291-301. doi: 10.1523/JNEUROSCI.3443-04.2004. J Neurosci. 2004. PMID: 15601935 Free PMC article.
-
Broadening the spectrum of actin-based protrusive activity mediated by Arp2/3 complex-facilitated polymerization: motility of cytoplasmic ridges and tubular projections.Cytoskeleton (Hoboken). 2014 Aug;71(8):484-500. doi: 10.1002/cm.21186. Epub 2014 Aug 26. Cytoskeleton (Hoboken). 2014. PMID: 25111797 Free PMC article.
References
-
- Beckerle MC. Spatial control of actin filament assembly: lessons from Listeria. Cell. 1998;95:741–748. - PubMed
-
- Berg HC, Block SM. A miniature flow cell designed for rapid exchange of media under high-power microscope objectives. J Gen Microbiol. 1984;130:2915–2920. - PubMed
-
- Bershadsky A, Chausovsky A, Becker E, Lyubimova A, Geiger B. Involvement of microtubules in the control of adhesion-dependent signal transduction. Curr Biol. 1996;6:1279–1289. - PubMed
-
- Bershadsky AD, Vaisberg EA, Vasiliev JM. Pseudopodial activity at the active edge of migrating fibroblast is decreased after drug-induced microtubule depolymerization. Cell Motil Cytoskeleton. 1991;19:152–158. - PubMed
-
- Best A, Ahmed S, Kozma R, Lim L. The Ras-related GTPase Rac1 binds tubulin. J Biol Chem. 1996;271:3756–3762. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

