Neoplastic transformation of RK3E by mutant beta-catenin requires deregulation of Tcf/Lef transcription but not activation of c-myc expression
- PMID: 10409758
- PMCID: PMC84421
- DOI: 10.1128/MCB.19.8.5696
Neoplastic transformation of RK3E by mutant beta-catenin requires deregulation of Tcf/Lef transcription but not activation of c-myc expression
Abstract
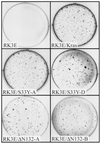
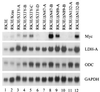
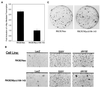
Current models predict that beta-catenin (beta-cat) functions in Wnt signaling via activation of Tcf/Lef target genes and that its abundance is regulated by the adenomatous polyposis coli (APC) and glycogen synthase kinase 3beta (GSK3beta) proteins. In colon and other cancers, mutations in APC or presumptive GSK3beta phosphorylation sites of beta-cat are associated with constitutive activation of Tcf/Lef transcription. In spite of assumptions about its oncogenic potential, prior efforts to demonstrate that mutated beta-cat will induce neoplastic transformation have yielded equivocal results. We report here that mutated, but not wild-type, beta-cat proteins induced neoplastic transformation of RK3E, an adenovirus E1A-immortalized epithelial cell line. Analysis of the properties of mutant beta-cat proteins and studies with a dominant negative Tcf-4 mutant indicated that the ability of beta-cat to bind and activate Tcf/Lef factors is crucial for transformation. c-myc has recently been implicated as a critical Tcf-regulated target gene. However, c-myc was not consistently activated in beta-cat-transformed RK3E cells, and a dominant negative c-Myc mutant protein failed to inhibit beta-cat transformation. Our findings underscore the role of beta-cat mutations and Tcf/Lef activation in cancer and illustrate a useful system for defining critical factors in beta-cat transformation.
Figures





Similar articles
-
gamma-catenin is regulated by the APC tumor suppressor and its oncogenic activity is distinct from that of beta-catenin.Genes Dev. 2000 Jun 1;14(11):1319-31. Genes Dev. 2000. PMID: 10837025 Free PMC article.
-
Beta- and gamma-catenin mutations, but not E-cadherin inactivation, underlie T-cell factor/lymphoid enhancer factor transcriptional deregulation in gastric and pancreatic cancer.Cell Growth Differ. 1999 Jun;10(6):369-76. Cell Growth Differ. 1999. PMID: 10392898
-
ITF-2, a downstream target of the Wnt/TCF pathway, is activated in human cancers with beta-catenin defects and promotes neoplastic transformation.Cancer Cell. 2002 Mar;1(2):145-55. doi: 10.1016/s1535-6108(02)00035-1. Cancer Cell. 2002. PMID: 12086873
-
Signaling through beta-catenin and Lef/Tcf.Cell Mol Life Sci. 1999 Oct 30;56(5-6):523-37. doi: 10.1007/s000180050449. Cell Mol Life Sci. 1999. PMID: 11212302 Free PMC article. Review.
-
The Yin-Yang of TCF/beta-catenin signaling.Adv Cancer Res. 2000;77:1-24. doi: 10.1016/s0065-230x(08)60783-6. Adv Cancer Res. 2000. PMID: 10549354 Review.
Cited by
-
Tumour suppressor TRIM33 targets nuclear β-catenin degradation.Nat Commun. 2015 Feb 2;6:6156. doi: 10.1038/ncomms7156. Nat Commun. 2015. PMID: 25639486 Free PMC article.
-
Wnt target gene activation requires β-catenin separation into biomolecular condensates.PLoS Biol. 2024 Sep 24;22(9):e3002368. doi: 10.1371/journal.pbio.3002368. eCollection 2024 Sep. PLoS Biol. 2024. PMID: 39316611 Free PMC article.
-
FOXQ1, a novel target of the Wnt pathway and a new marker for activation of Wnt signaling in solid tumors.PLoS One. 2013;8(3):e60051. doi: 10.1371/journal.pone.0060051. Epub 2013 Mar 26. PLoS One. 2013. PMID: 23555880 Free PMC article.
-
Cinobufagin Suppresses Melanoma Cell Growth by Inhibiting LEF1.Int J Mol Sci. 2020 Sep 13;21(18):6706. doi: 10.3390/ijms21186706. Int J Mol Sci. 2020. PMID: 32933177 Free PMC article.
-
Current understanding and dispute on the function of the Wnt signaling pathway effector TCF7L2 in hepatic gluconeogenesis.Genes Dis. 2015 Nov 17;3(1):48-55. doi: 10.1016/j.gendis.2015.10.002. eCollection 2016 Mar. Genes Dis. 2015. PMID: 30258876 Free PMC article. Review.
References
-
- Aberle H, Butz S, Stappert J, Weissig H, Kemler R, Hoschuetzky H. Assembly of the cadherin-catenin complex in vitro with recombinant proteins. J Cell Sci. 1994;107:3655–3663. - PubMed
-
- Barth A I, Nathke I S, Nelson W J. Cadherins, catenins and APC protein: interplay between cytoskeletal complexes and signaling pathways. Curr Opin Cell Biol. 1997;9:683–690. - PubMed
-
- Behrens J, von Kries J P, Kuhl M, Bruhn L, Wedlich D, Grosschedl R, Birchmeier W. Functional interaction of β-catenin with the transcription factor LEF-1. Nature. 1996;382:638–642. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
