Mammalian unfolded protein response inhibits cyclin D1 translation and cell-cycle progression
- PMID: 10411905
- PMCID: PMC17546
- DOI: 10.1073/pnas.96.15.8505
Mammalian unfolded protein response inhibits cyclin D1 translation and cell-cycle progression
Abstract
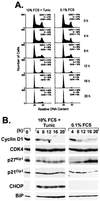
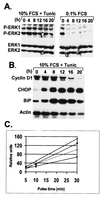
Alterations in normal protein biogenesis and the resulting accumulation of improperly folded proteins in the endoplasmic reticulum (ER) trigger a stress response that up-regulates the expression of ER chaperones, while coordinately repressing overall protein synthesis and causing cell-cycle arrest. Activation of this unfolded protein response (UPR) in mouse NIH 3T3 fibroblasts with the glycosylation inhibitor tunicamycin led to a decline in cyclin D- and E-dependent kinase activities and to G(1) phase arrest. Cyclin D1 protein synthesis was rapidly inhibited by tunicamycin treatment. However, the drug did not significantly affect the mitogen-dependent activities of the extracellular signal-activated protein kinases ERK1 and ERK2 or the level of cyclin D1 mRNA until much later in the response. Therefore, the UPR triggers a signaling pathway that blocks cyclin D1 translation despite continuous mitogenic stimulation. Enforced overexpression of cyclin D1 in tunicamycin-treated cells maintained cyclin D- and E-dependent kinase activities and kept cells in cycle in the face of a fully activated UPR. Translational regulation of cyclin D1 in response to ER stress is a mechanism for checkpoint control that prevents cell-cycle progression until homeostasis is restored.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

