Contributions of residual calcium to fast synaptic transmission
- PMID: 10414955
- PMCID: PMC6782810
- DOI: 10.1523/JNEUROSCI.19-15-06257.1999
Contributions of residual calcium to fast synaptic transmission
Abstract
Fast neurotransmitter release is driven by high calcium (10-100 microM) near open channels (Ca(local)), followed by a much smaller (<1 microM), longer-lasting residual calcium (Ca(res)). The most prominent component of release, phasic release, lasts several milliseconds and is thought to be triggered by Ca(local). A transient tail of release then continues over the next 20 msec at 1-10% of peak rates. This transient component of release, which we refer to as TR, is poorly understood, and there is conflicting evidence regarding the role of Ca(local) and Ca(res) in its generation. We used optical methods to monitor Ca(res) and whole-cell voltage-clamp recordings to study TR at synapses between granule cells and stellate cells in rat cerebellar slices. After stimulation the probability of release is elevated greatly, peaking at 500 microseconds and then slowly declining to prestimulus levels after tens of milliseconds. After speeding the decay of Ca(res) levels with EGTA, release is confined to a 3 msec interval, and TR is eliminated. Thus, we find that Ca(res) accounts for a transient tail of release on the millisecond time scale that helps to shape the average synaptic current and accounts for at least 20% of the synaptic charge in the 20 msec interval after stimulation. Ca(res)-dependent TR is likely to contribute significantly to fast synaptic transmission under physiological conditions, particularly during high-frequency bursts that elevate Ca(res).
Figures


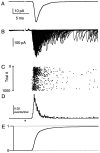





References
-
- Augustine GJ, Adler EM, Charlton MP. The calcium signal for transmitter secretion from presynaptic nerve terminals. In: Stanley EF, Nowycky MC, Triggle DJ, editors. Calcium entry and action at the presynaptic nerve terminal. New York Academy of Sciences; New York: 1991. pp. 365–381. - PubMed
-
- Barbour B, Keller BU, Llano I, Marty A. Prolonged presence of glutamate during excitatory synaptic transmission to cerebellar Purkinje cells. Neuron. 1994;12:1331–1343. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
