Characterization of integrin-tetraspanin adhesion complexes: role of tetraspanins in integrin signaling
- PMID: 10427099
- PMCID: PMC2156181
- DOI: 10.1083/jcb.146.2.477
Characterization of integrin-tetraspanin adhesion complexes: role of tetraspanins in integrin signaling
Abstract
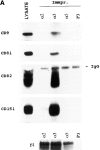
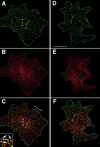
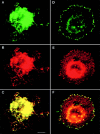
Tetraspanins (or proteins from the transmembrane 4 superfamily, TM4SF) form membrane complexes with integrin receptors and are implicated in integrin-mediated cell migration. Here we characterized cellular localization, structural composition, and signaling properties of alpha3beta1-TM4SF adhesion complexes. Double-immunofluorescence staining showed that various TM4SF proteins, including CD9, CD63, CD81, CD82, and CD151 are colocalized within dot-like structures that are particularly abundant at the cell periphery. Differential extraction in conjunction with chemical cross-linking indicated that the cell surface fraction of alpha3beta1-TM4SF protein complexes may not be directly linked to the cytoskeleton. However, in cells treated with cytochalasin B alpha3beta1-TM4SF protein complexes are relocated into intracellular vesicles suggesting that actin cytoskeleton plays an important role in the distribution of tetraspanins into adhesion structures. Talin and MARCKS are partially codistributed with TM4SF proteins, whereas vinculin is not detected within the tetraspanin-containing adhesion structures. Attachment of serum-starved cells to the immobilized anti-TM4SF mAbs induced dephosphorylation of focal adhesion kinase (FAK). On the other hand, clustering of tetraspanins in cells attached to collagen enhanced tyrosine phosphorylation of FAK. Furthermore, ectopic expression of CD9 in fibrosarcoma cells affected adhesion-induced tyrosine phosphorylation of FAK, that correlated with the reorganization of the cortical actin cytoskeleton. These results show that tetraspanins can modulate integrin signaling, and point to a mechanism by which TM4SF proteins regulate cell motility.
Figures












References
-
- Barnstable C.J., Bodmer W.F., Brown G., Galfré G., Milstein C., Williams A.F., Zeigler A. Production of monoclonal antibodies to group A erythrocytes, HLA and other human cell surface antigens—new tools for genetic analysis. Cell. 1978;14:9–20. - PubMed
-
- Berditchevski F., Bazzoni G., Hemler M.E. Specific association of CD63 with the VLA-3 and VLA-6 integrins. J. Biol. Chem. 1995;270:17784–17790. - PubMed
-
- Berditchevski F., Chang S., Bodorova J., Hemler M.E. Generation of monoclonal antibodies to integrin-associated proteins. Evidence that α3β1 complexes with EMMPRIN/basigin/OX47/M6 J. Biol. Chem. 272 1997. 29174 29180a - PubMed
-
- Berditchevski F., Tolias K.F., Wong K., Carpenter C.L., Hemler M.E. Novel link between integrins, TM4SF proteins (CD63, CD81) and phosphatidylinositol 4-kinase J. Biol. Chem. 272 1997. 2595 2598b - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

