Ovarian hormone dependence of alpha(1)-adrenoceptor activation of the nitric oxide-cGMP pathway: relevance for hormonal facilitation of lordosis behavior
- PMID: 10436072
- PMCID: PMC6782854
- DOI: 10.1523/JNEUROSCI.19-16-07191.1999
Ovarian hormone dependence of alpha(1)-adrenoceptor activation of the nitric oxide-cGMP pathway: relevance for hormonal facilitation of lordosis behavior
Abstract
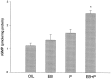
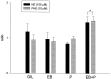
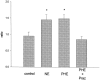
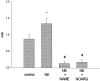
The ovarian hormones estradiol (E(2)) and progesterone (P) facilitate rat lordosis behavior in part by regulating the expression of and signal transduction by adrenoceptors in the hypothalamus (HYP) and preoptic area (POA). The major adrenoceptor subtype mediating E(2) and P facilitation of lordosis is the alpha(1)-adrenoceptor. In the present studies, we tested the hypotheses that (1) alpha(1)-adrenoceptors in the HYP enhance lordosis responses by activating the nitric oxide (NO)-cGMP signaling pathway, and (2) coupling of alpha(1)-adrenoceptors to this signal transduction pathway is hormone-dependent. Basal levels of cGMP were significantly higher in HYP and POA slices from animals treated with E(2) and P when compared with slices from ovariectomized controls or females treated with only E(2) or P. When slices of HYP and POA from ovariectomized female rats were incubated with norepinephrine or the selective alpha(1)-adrenoceptor agonist phenylephrine, cGMP accumulation was observed only if slices had been derived from females treated with both E(2) and P before experimentation. Moreover, alpha(1)-adrenoceptor stimulation of cGMP synthesis was blocked by an inhibitor of NO synthase, confirming that these receptors act by NO-mediated stimulation of soluble guanylyl cyclase. Behavioral studies demonstrated further that the cell-permeable cGMP analog 8-bromoadenosine-cGMP reverses the inhibitory effects of the alpha(1)-adrenoceptor antagonist prazosin on lordosis behavior in E(2)- and P-treated female rats. Thus, the NO-cGMP pathway mediates the facilitatory effects of alpha(1)-adrenoceptors on lordosis behavior in female rats, and previous exposure of the HYP and POA to both E(2) and P are required to link alpha(1)-adrenoceptors to this pathway.
Figures
Similar articles
-
Functional interactions between estrogen and insulin-like growth factor-I in the regulation of alpha 1B-adrenoceptors and female reproductive function.J Neurosci. 2002 Mar 15;22(6):2401-8. doi: 10.1523/JNEUROSCI.22-06-02401.2002. J Neurosci. 2002. PMID: 11896179 Free PMC article.
-
Estradiol and progesterone modulate the nitric oxide/cyclic gmp pathway in the hypothalamus of female rats and in GT1-1 cells.Endocrine. 2004 Jul;24(2):177-84. doi: 10.1385/ENDO:24:2:177. Endocrine. 2004. PMID: 15347845
-
Evidence that oestradiol attenuates beta-adrenoceptor function in the hypothalamus of female rats by altering receptor phosphorylation and sequestration.J Neuroendocrinol. 2000 Nov;12(11):1060-6. doi: 10.1046/j.1365-2826.2000.00562.x. J Neuroendocrinol. 2000. PMID: 11069121
-
Mechanisms of ovarian steroid regulation of norepinephrine receptor-mediated signal transduction in the hypothalamus: implications for female reproductive physiology.Horm Behav. 2001 Sep;40(2):169-77. doi: 10.1006/hbeh.2001.1676. Horm Behav. 2001. PMID: 11534978 Review.
-
Ovarian steroid and growth factor regulation of female reproductive function involves modification of hypothalamic alpha 1-adrenoceptor signaling.Ann N Y Acad Sci. 2003 Dec;1007:153-61. doi: 10.1196/annals.1286.015. Ann N Y Acad Sci. 2003. PMID: 14993049 Review.
Cited by
-
Functional interactions between estrogen and insulin-like growth factor-I in the regulation of alpha 1B-adrenoceptors and female reproductive function.J Neurosci. 2002 Mar 15;22(6):2401-8. doi: 10.1523/JNEUROSCI.22-06-02401.2002. J Neurosci. 2002. PMID: 11896179 Free PMC article.
-
Methamphetamine-enhanced female sexual motivation is dependent on dopamine and progesterone signaling in the medial amygdala.Horm Behav. 2015 Jan;67:1-11. doi: 10.1016/j.yhbeh.2014.10.004. Epub 2014 Nov 11. Horm Behav. 2015. PMID: 25448531 Free PMC article.
-
Estrogen regulation of proteins in the rat ventromedial nucleus of the hypothalamus.J Proteome Res. 2008 Nov;7(11):5040-8. doi: 10.1021/pr8005974. Epub 2008 Oct 8. J Proteome Res. 2008. PMID: 18841879 Free PMC article.
-
Facilitation of estrous behavior by vaginal cervical stimulation in female rats involves alpha1-adrenergic receptor activation of the nitric oxide pathway.Behav Brain Res. 2007 Jan 25;176(2):237-43. doi: 10.1016/j.bbr.2006.10.007. Epub 2006 Nov 13. Behav Brain Res. 2007. PMID: 17095102 Free PMC article.
-
Estradiol and progesterone modulate the nitric oxide/cyclic gmp pathway in the hypothalamus of female rats and in GT1-1 cells.Endocrine. 2004 Jul;24(2):177-84. doi: 10.1385/ENDO:24:2:177. Endocrine. 2004. PMID: 15347845
References
-
- Ari IL, Schwarz L, Atlas D. Cholinergic-induced [3H] noradrenaline release in rat brain cortical slices is mediated via a pertussis toxin sensitive GTP binding protein and involves activation of protein kinase C. Cell Signal. 1989;1:461–470. - PubMed
-
- Beyer C, Gonzalez-Flores O, Gonzalez-Mariscal G. Progesterone receptor participates in the stimulatory effect of LHRH, prostaglandin E2, and cyclic AMP on lordosis and proceptive behaviours in rats. J Neuroendocrinol. 1997;9:609–614. - PubMed
-
- Bhat GK, Mahesh VB, Lamar CA, Ping L, Aguan K, Brann DW. Histochemical localization of nitric oxide neurons in the hypothalamus: association with gonadotropin-releasing hormone neurons and co-localization with N-methyl-d-aspartate receptors. Neuroendocrinology. 1995;62:187–197. - PubMed
-
- Bonavera JJ, Sahu A, Kalra PS, Kalra SP. Evidence that nitric oxide may mediate the ovarian steroid-induced luteinizing hormone surge: involvement of excitatory amino acids. Endocrinology. 1993;133:2481–2487. - PubMed
-
- Brann DW, Bhat GK, Lamar CA, Mahesh VB. Gaseous transmitters and neuroendocrine regulation. Neuroendocrinology. 1997;65:385–395. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
