Interleukin-3 and granulocyte-macrophage colony-stimulating factor enhance the generation and function of dendritic cells
- PMID: 10447725
- PMCID: PMC2326801
- DOI: 10.1046/j.1365-2567.1999.00741.x
Interleukin-3 and granulocyte-macrophage colony-stimulating factor enhance the generation and function of dendritic cells
Abstract
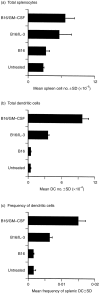
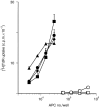
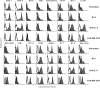
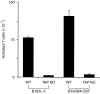
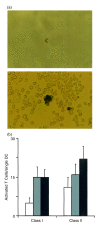
Dendritic cells, well-known for their potent antigen-presenting activity, are generally present at very low frequency in the spleens of naive mice. We examined the ability of mice to generate functional dendritic cells (DC) following exposure to the cytokines interleukin-3 (IL-3) and granulocyte-macrophage colony-stimulating factor (GM-CSF). Tumours secreting these cytokines provided a continuous stimulus resulting in a greatly increased number and frequency of DC in the spleen. These cells were purified by conventional DC isolation techniques and were found to exhibit many of the characteristics of DC from unmanipulated mice, including high allo-stimulatory activity in mixed lymphocyte reactions and expression of many similar cell surface markers. Using ovalbumin-peptide specific class I- and class II-restricted hybridomas containing the lacZ reporter gene, we found that these cytokine-generated DC had a greatly increased efficacy in the uptake and processing of particulate antigen. These cells appear to have retained the ability to ingest antigen that is generally associated with immature DC, but also exhibit the peptide/major histocompatibility complex (MHC)-presenting capabilities of mature DC. Development of an assay to measure the activity of a single DC revealed that these dual activities were the properties of the majority of the cytokine-generated DC. These findings indicate that exposure in vivo to the cytokines IL-3 and GM-CSF can result in the generation of large numbers of DC with increased capability of stimulating T cells. Thus, these cells may be important in vivo in the process of cross-priming and the subsequent generation of tumour-reactive cytotoxic T lymphocytes (CTL).
Figures

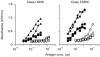
Similar articles
-
Therapy of established tumour with a hybrid cellular vaccine generated by using granulocyte-macrophage colony-stimulating factor genetically modified dendritic cells.Immunology. 1999 Aug;97(4):616-25. doi: 10.1046/j.1365-2567.1999.00823.x. Immunology. 1999. PMID: 10457215 Free PMC article.
-
Comparison of the functional properties of murine dendritic cells generated in vivo with Flt3 ligand, GM-CSF and Flt3 ligand plus GM-SCF.Cytokine. 2002 Feb 7;17(3):119-30. doi: 10.1006/cyto.2001.0995. Cytokine. 2002. PMID: 11895330
-
Immunophenotypical and functional heterogeneity of dendritic cells generated from murine bone marrow cultured with different cytokine combinations: implications for anti-tumoral cell therapy.Immunology. 1999 Apr;96(4):569-77. doi: 10.1046/j.1365-2567.1999.00728.x. Immunology. 1999. PMID: 10233743 Free PMC article.
-
Cytokines in the generation and maturation of dendritic cells: recent advances.Eur Cytokine Netw. 2002 Apr-Jun;13(2):186-99. Eur Cytokine Netw. 2002. PMID: 12101074 Review.
-
Cytokines amplify the function of accessory cells.Immunol Lett. 1988 Mar;17(3):197-202. doi: 10.1016/0165-2478(88)90028-4. Immunol Lett. 1988. PMID: 3286486 Review.
Cited by
-
Genetic deletion of mPGES-1 abolishes PGE2 production in murine dendritic cells and alters the cytokine profile, but does not affect maturation or migration.Prostaglandins Leukot Essent Fatty Acids. 2011 Mar-Apr;84(3-4):113-21. doi: 10.1016/j.plefa.2010.10.003. Epub 2010 Dec 28. Prostaglandins Leukot Essent Fatty Acids. 2011. PMID: 21190819 Free PMC article.
-
Identification of T-cell epitopes in Francisella tularensis using an ordered protein array of serological targets.Immunology. 2011 Mar;132(3):348-60. doi: 10.1111/j.1365-2567.2010.03387.x. Epub 2011 Jan 7. Immunology. 2011. PMID: 21214540 Free PMC article.
-
IL-3 promotes the development of experimental autoimmune encephalitis.JCI Insight. 2016 Oct 6;1(16):e87157. doi: 10.1172/jci.insight.87157. JCI Insight. 2016. PMID: 27734026 Free PMC article.
-
Recombinant murine granulocyte-macrophage colony-stimulating factor modulates the course of pulmonary histoplasmosis in immunocompetent and immunodeficient mice.Antimicrob Agents Chemother. 2000 Dec;44(12):3328-36. doi: 10.1128/AAC.44.12.3328-3336.2000. Antimicrob Agents Chemother. 2000. PMID: 11083636 Free PMC article.
-
IL-3 contributes to development of lupus nephritis in MRL/lpr mice.Kidney Int. 2015 Nov;88(5):1088-98. doi: 10.1038/ki.2015.196. Epub 2015 Jul 1. Kidney Int. 2015. PMID: 26131743
References
-
- Pierre P, Turley SJ, Gatti E, et al. Developmental regulation of MHC class II transport in mouse dendritic cells. Nature. 1997;388:787. - PubMed
-
- Cella M, Engering A, Pinet V, Pieters J, Lanzavecchia A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature. 1997;388:782. - PubMed
-
- Brodsky FM, Guagliardi LE. The cell biology of antigen processing and presentation. Ann Rev Immunol. 1991;9:707. - PubMed
-
- Monaco JJ. Pathways for the processing and presentation of antigens to T cells. J Leukocyte Biol. 1995;57:543. - PubMed
-
- Braciale TJ, Braciale VL. Viral antigen presentation and MHC assembly. Semin Immunol. 1992;4:81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

