Approach to withdrawal from tacrolimus in a fully allogeneic murine skin graft model
- PMID: 10447745
- PMCID: PMC2326833
- DOI: 10.1046/j.1365-2567.1999.00760.x
Approach to withdrawal from tacrolimus in a fully allogeneic murine skin graft model
Abstract
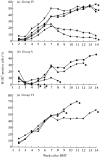
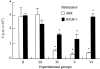
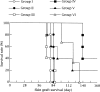
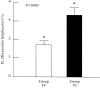
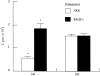
With few exceptions, transplant patients must take immunosuppressants throughout their lives. In this study, we used anti-T-cell receptor (TCR/CD3) monoclonal antibodies (mAbs) to induce immunological tolerance to alloantigens after withdrawal from tacrolimus in a fully allogeneic murine skin graft model. Skin grafts from AKR donor mice were maintained in C57BL/6 recipients by administering tacrolimus for one month. Anti-T-cell receptor (TCR) alphabeta mAb was administered to recipient mice on the day of withdrawal from tacrolimus administration. Seven days after mAb administration, the recipient mice were treated with various combinations of the following treatments: low-dose whole body irradiation, AKR bone marrow transfer (BMT), and anti-CD3 mAb administration. The control recipient mice did not receive treatment with either mAb, nor any other treatment. All the control recipient mice showed rejection of AKR skin grafts 42 days after tacrolimus withdrawal (mean skin graft survival: 77 days). Mice treated with a combination of anti-TCR alphabeta antibody, low-dose irradiation and AKR BMT showed stable chimerism in their peripheral blood lymphocytes and significantly prolonged skin graft survival (mean skin graft survival: >151.2+/-15.3 days). Mice given the combination of anti-TCR alphabeta mAb, anti-CD3 mAb, low-dose irradiation, and AKR BMT exhibited more stable chimerism but had earlier skin graft rejection (mean skin graft survival: 116.7+/-17.6 days) than the mice that did not receive anti-CD3 mAb. These results suggest that anti-TCR alphabeta mAb, but not anti-CD3 mAb, in combination with low-dose irradiation and BMT, is useful for long-lasting allograft survival after withdrawal from tacrolimus in mice with fully allogeneic skin grafts.
Figures

Similar articles
-
Additional monoclonal antibody (mAB) injections can replace thymic irradiation to allow induction of mixed chimerism and tolerance in mice receiving bone marrow transplantation after conditioning with anti-T cell mABs and 3-Gy whole body irradiation.Transplantation. 1996 Feb 15;61(3):469-77. doi: 10.1097/00007890-199602150-00027. Transplantation. 1996. PMID: 8610363
-
Tolerance induction in a fully allogeneic combination using anti-T cell receptor-alpha beta monoclonal antibody, low dose irradiation, and donor bone marrow transfusion.Transplantation. 1995 Feb 15;59(3):395-401. Transplantation. 1995. PMID: 7871570
-
Combined effects of calcineurin inhibitors or sirolimus with anti-CD40L mAb on alloengraftment under nonmyeloablative conditions.Blood. 2002 Nov 1;100(9):3400-7. doi: 10.1182/blood-2002-03-0872. Blood. 2002. PMID: 12384443
-
Prolongation of allogeneic skin graft survival by injection of anti-Ly49A monoclonal antibody YE1/48.Clin Immunol. 2003 Feb;106(2):148-54. doi: 10.1016/s1521-6616(02)00041-4. Clin Immunol. 2003. PMID: 12672405
-
Simultaneous bone marrow and composite tissue transplantation in rats treated with nonmyeloablative conditioning promotes tolerance.Transplantation. 2013 Jan 27;95(2):301-8. doi: 10.1097/TP.0b013e31827899fc. Transplantation. 2013. PMID: 23250336 Free PMC article.
References
-
- Thomson AW, Woo J. Immunosuppressive properties of FK-506 and rapamycin. Lancet. 1989;2:443. - PubMed
-
- Sigal NH, Dummont FJ. Cyclosporine A, FK-506, and rapamycin: pharmacologic probes of lymphocyte signal transduction. Annu Rev Immunol. 1992;10:519. - PubMed
-
- Ho M, Wajszczuk CP, Hardy A, et al. Infections in kidney, heart and liver transplant recipients on cyclosporine. Transplant Proc. 1983;15(Suppl. 1):2768. - PubMed
-
- Blohmé I, Brynger H. Malignant disease in renal transplant patients. Transplantation. 1985;39:23. - PubMed
-
- Penn I. Cancers following cyclosporine therapy. Transplantation. 1987;43:32. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

