Immune responses to Plasmodium falciparum-merozoite surface protein 1 (MSP1) antigen, II. Induction of parasite-specific immunoglobulin G in unsensitized human B cells after in vitro T-cell priming with MSP119
- PMID: 10447773
- PMCID: PMC2326866
- DOI: 10.1046/j.1365-2567.1999.00804.x
Immune responses to Plasmodium falciparum-merozoite surface protein 1 (MSP1) antigen, II. Induction of parasite-specific immunoglobulin G in unsensitized human B cells after in vitro T-cell priming with MSP119
Abstract
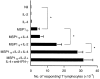
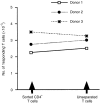
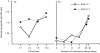
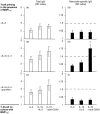
A baculovirus recombinant antigen corresponding to the C-terminal 19 000 MW fragment of Plasmodium falciparum merozoite surface protein 1 (MSP119), has been used to prime T cells from individuals with no previous exposure to malaria, to provide help for the induction of a parasite specific antibody response in vitro. Although MSP119 alone could induce a small but detectable T-cell response, which included interleukin-4 (IL-4) secretion, this response was significantly increased by the presence of IL-2. In addition, IL-4 was shown to synergize with IL-2 for the induction of antigen-specific T-cell responses. If interferon-gamma (IFN-gamma), IL-12, or neutralizing anti-IL-4 antibody was present at the time of priming, the T-cell responses were abolished. Parasite-specific immunoglobulin G (IgG) could be detected after secondary restimulation with MSP119, IL-10 and anti-CD40 monoclonal antibody in cultures containing MSP119 primed T cells, autologous B cells, IL-2 and IL-4. No antibody was secreted in the absence of primed T cells in this B-cell culture assay. These data show that recombinant MSP119, a leading malaria vaccine candidate, can prime non-immune human lymphocytes under defined in vitro experimental conditions, which include regulatory cytokines and/or other costimulatory molecules. This is a complementary approach for exploring immunogenic mechanisms of potential vaccine candidates such as P. falciparum antigens in humans.
Figures

References
-
- Engers HD, Godal T. Malaria vaccine development. Parasitol Today. 1998;14:56. - PubMed
-
- Good MF, Kaslow DC, Miller LH. Pathways and strategies for developing a malaria blood-stage vaccine. Annu Rev Immunol. 1998;16:57. - PubMed
-
- Perraut R, Garraud O. Essais vaccinaux et réponse immune contre Plasmodium falciparum chez le singe. Saimiri Méd Trop. 1998;58:76. - PubMed
-
- Holder AA, Riley EM. Human immune response to MSP-1. Parasitol Today. 1996;12:173. - PubMed
-
- Egan AF, Morris J, Barnish G, et al. Clinical immunity to Plasmodium falciparum malaria is associated with serum antibodies to three 19-kDa C-terminal fragment of the merozoite surface antigen, PfMSP-1. J Infect Dis. 1996;173:765. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

